DIT review - Heme 2 Flashcards
Where does erythropoeisis occur in early life and later life
- Fetal development - liver
- After 28 weeks - bone marrow
- Infancy and childhood:
- Flat bones
- Sternum, pelvis, ribs, cranial bones, vertebrae, long bones of leg
- Later adolescence and adulthood
- Axial skeleton
- Vertebrae, sternum, ribs, and pelvis
- Infancy and childhood:
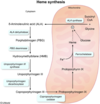
What is the rate limiting enzyme of heme synthesis
ALA synthase:
Succinyl CoA + Glycine –> ALA
What is the cofactor required for ALA synthase
B6
What are causes of polycythemia (increased RBC)
- Polycythemia vera – monoclonal proliferation of red cells
- Chronic hypoxia – need to increase O2 carrying capacity so kidney produces more erythropoietin to make more RBCs
- Pulmonary disease
- Cyanotic heart disease
- High altitudes
- Inappropriate elevation of EPO – e.g. EPO-producing tumor
- Trisomy 21
What are the EPO-producing tumors?
THINK: Potentially Really High Hematocrit
- Pheochromocytoma
- Renal cell carcinoma
- Hepatocellular carcinoma
- Hemangioblastoma
What is the enzyme deficiency in Acute intermittent porphyria
Porphobilinogen (PBG) deaminase
THINK:
o Acute intermittent = guys hollering “damn” (deam-inase) intermittently at A CUTE pretty big girl (PBG)

Presentation of acute intermittent porphyria
§ Symptoms – 5 P’s
· Painful abdomen
· Port wine colored urine (due to increase PGB)
· Polyneuropathy
· Psychological disturbances
· Precipitated by drugs (CYP450 inducers – e.g. Rifampin), alcohol, and starvation
Treatment of acute intermittent porphyria
· Glucose + heme = inhibition of ALA synthase
What is the deficienct enzyme in porphyria cutanea tarda?
Uroporphyrinogen carboxylase
THINK of a homeless man living in a cardboard box (carbox)
Presentatin of porphyria cutanea tarda
§ Symptoms
· Blistering cutaneous photosensitivity
· Hyperpigmentation
· Hyerptrichosis (extra hair)
· Tea colored urine
· Exacerbated with alcohol consumption
· Associated with Hepatitis C
§ (THINK of a stereotypical homeless man)
· Alcoholic, face blistered and dark from sun, facial hair, liver disease
Enzyme defiecient in lead poisoning
ALA dehydratase and Ferrochelatase

Presentation of lead poisoning
- GI (abd pain, constipation, anorexia)
- Neuro (cognitive defects, peripheral neuropathy, encephalopathy, memory loss, delirium)
- Hematologic (microcytic anemia with basophilic stippling, ringed sideroblasts in marrow)
- Burton lines – lead lines in gingiva and gums
- Hyper dense lines on metaphysis of long bones
- Renal failure
What are the different causes of microcytic anemia
Iron deficiency
Anemia of chronic disease (late)
Thalassemia
Lead poisoning
Sideroblastic anemia
What is the basic principle behing microcytic anemia
- MCV < 80
- Due to decreased production of hemoglobin = extra division to maintain Hb concentration
- Hemoglobin = heme + globin
- Heme = iron + protoporphyrin
- Hemoglobin = heme + globin
- Cells are small and hypochromic (pale)
What are lab values of iron deficiency anemia (ferritin, serum iron, TIBC, % saturation)
- Low ferritin (low iron stores)
- Low serum iron
- Low % saturation (of transferrin – iron transporter)
- High TIBC (Total iron-binding capacity = # of transferrin molecules in the blood – will be elevated because the liver is pumping out more in a state of low iron in order to replenish iron
- Ferritin and TIBC are always opposite
What is the molecule responsible for anemia of chronic disease
Hepcidin sequesters iron into storage sites
What are lab values of anemia of chronic disease(ferritin, serum iron, TIBC, % saturation)
- Labs:
- High ferritin
- Low TIBC
- Low serum iron
- Low % saturation
- Early disease presents as nonhemolytic, normocytic anemia
- Late disease presents as microcytic anemia
Describe the defect in alpha thalassemia
- Defect in a-globin synthesis
- Alpha Thalassemia
- Due to alpha-globin gene deletion = decreased alpha-globin synthesis
- There are 4 alpha genes on chromosome 16
Describe the different types of alpha-thalassemia
- 1 gene deleted = asymptomatic
- 2 genes deleted = mild anemia
- Cis = increased risk in offspring – Asians
- Trans = Africans
- 3 genes deleted
- Beta chains form tetramers – B4 = Hemoglobin H (HbH)
- 4 genes deleted
- No a-globin at all
- Gamma (y) chains form tetramer – y4 = Hemoglobin Barts (Hb Barts)
- Incompatible with life – hydrops fetalis
Describe the defect in beta thalassemia
- Due to beta globin gene mutation
- There are 2 beta genes on chromosome 11
- Mutations can result in absent (B0) or diminished (B+) production of b-globin
- Seen in Mediterranean populations
Describe beta-thalassemia minor (genes, presentation, types of Hb, histology)
- B-thalassemia minor (B/B+)
- Decreased amount of B-globin
- Minimal anemia
- Increased HbA2 (a2d2) and HbF (a2y2)
- Will see target cells
Describe beta thalassemia major (genes, presentation, histology)
- B-thalassemia major (B0/B0)
- No B-globin at all
- Severe anemia requiring blood transfusions
- Risk of hemochromatosis
- High HbF at birth is temporarily protective
- Will see target cells
- Erythroid hyperplasia – hematopoiesis occurring in unusual places, such as face, skull, liver, spleen
- “Crewcut” appearance on X-ray (“hair-on-end”)
- “Chipmunk” facies
- Hepatosplenomegaly
- Risk of aplastic crisis with Parvovirus B19 infection of erythroid precursors

Describe the defect and causes of sideroblastic anemia
- Defect in heme synthesis
- Defect in protoporphyrin synthesis leads to iron buildup in the mitochondria = iron-laden mitochondria form a ring around nucleus of erythroid precursors
- Causes:
- Congenital:
- ALA synthetase deficiency
- Acquired:
- Lead poisoning
- Vitamin B6 deficiency (cofactor for ALA synthetase)
- Alcohol
- Congenital:
What are the lab values of sideroblastic anemia (ferritin, TIBC, serum iron, % saturation)
- High ferritin
- Low TIBC
- High serum iron
- High % saturation



