method used to determine the enthalpy of combustion
measure 100cm3 of water in beaker
weigh a spirit burner contiaining liquid to be burnt
measure initial temp of water using thermometer
use spirit burner to heat water
stop heating after a reasonable temp rise, stir and measure final temp of water
reweigh spirit burner
calculate temp change and heat energy change using q = mcΔt
calculate the mass of fuel burnt by subtraction and find number of moles of fuel using mass/Mr
calculate enthalpy change per mole of fuel used
why is value for enthalpy of combustion less exothermic than theoretical values?
heat loss to surroundings from spirit burner and calorimeter
loss of fuel from burner or water by evaporation
some heat used to raise temp of calorimeter
reaction unlikely to occur under standard conditions
ways to improve calorimetry experiment
using draught shield to reduce heat loss
add lid on calorimeter
minimise distance between flame and calorimeter
insulating calorimeter/spirit burner
burn in supply of pure oxgen to minimise incomplete combustion
method to determine enthalpy change of neutralisation
place polysstyrene cup in glass beaker
rinse measuring cylinder with 1 moldm-3 acid then measure 25cm3 and transfer to polystyrene cup
stir acid with themometer and record temp
rinse second measuring cylinder with 1 moldm-3 base and measure 25cm3
add base to acid and record highest temo reached
calculate temp change and heat energy change using q=mcΔt
calculate the amount of acid used, the moles of water formed and the enthalpy of neutralisation
test for Mg2+
dissolve compound in water to form aqueous solution
add a few drop of NaOH (aq) at first then excess NaOH
positive result = white ppt forms
test for hydroxide ions
warm with solid ammonium salt and test gas formed with red litmus paper or glass rod dipped in conc HCl(aq)
pungent ammonia gas released which changes red litmus paper to blue (as ammonia gas is alkaline) or produce white fumes with conc HCl
reaction : OH- + NH4+ → NH3 + H2O
test for sulfate ions
reagent = acidified barium chloride solution
white ppt forms
test for carbonate ions
add dilute HNO3
test gas with limewater
effervesence and turns colourless limewater cloudy
why is reflux often necessary involvign reactions with organic compound
organic compounds contain strong covalent bonds so organic reactions are slow
reflux increases the rate of reaction
why are reactants added slowly to the reaction flask with cooling before reflux ?
the reaction is often exothermic so this prevents temp of reaction mixture from rising too rapidly and dangerously splashing and forming unwanted side-products
purpose of condensor jacket in reflux apparatus
prevents vapour from escaping so the reactants can be boiled for long time without any loss of vapour
why are anti-bumping granules used in simple distillation
ensure smooth boiling because liquid can splash over into the condesnor causing impure product or can cause an explosion
what makes tertiary alcohols resistant to oxidation with acidified potassium dichromate
lack of H atom bonded to the C with the OH group attached
test for esters
warm with ethanol and add a few drops of conc sulphuric acid and smell mixture
sweet smell if ester present
test for carboxylic acids
sodium carbonate or Mg to test form phenol as not acidic enough to react with carbonates
effervescence and turns colourless limewater cloudy
effervescence - pop is heard when lighted splin is applied to gas produced (Hydrogen gas formed with Mg )
purification of organism crude product
contamination occurs due to byproducts or unreacted reactant
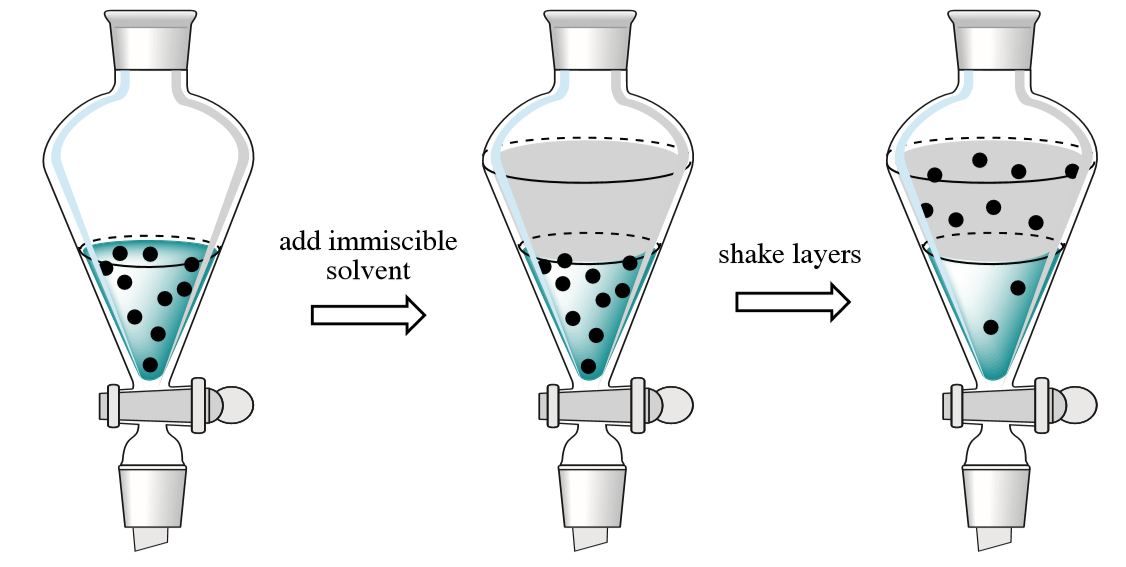
solvent extraction used to purify product:
stopper and shake organic liquid with an aqueous solution (usually sodium hydrogen carbonate)
impurities more soluble in aqueous solution than in organic liquid so move into the aq sol
organic liquid and aq sol are immiscible so form separate layers
remove stopper and open tap to run off bottom layer into beaker
close tap partially when bottom layer nearly drained to slow down flow
open tap again and run off second layer into second beaker
discard aqueous solution
repeat process using a new sample of aqueous sol