What are the 3 main types of bonds in chemistry?
Ionic, covalent and metallic
What are the particles involved in ionic bonding?
- A metal and a non metal ion.
- The particles are oppositely charged ions.
What are the particles involved in covalent bonding?
- Non metal particles
- The particles are atoms which share pairs of electrons.
What are the particles involved in metallic bonding?
- Metallic atoms.
- They share delocalised electrons.
Where do ionic bonds form?
In compounds formed from metals combined with non-metals.
Where do covalent bonds form?
In most non-metallic elements and in compounds of non-metals.
Where do metallic bonds form?
- Metallic elements
- Alloys.
What are electrostatic forces?
- Attraction or repulsion forces between particles that are caused by their electric charges.
- Between positive and negative ions
Does ionic bonding involve transfer or sharing of electrons?
Transfer
Does covalent bonding involve transfer or sharing of electrons?
Sharing “Co …” = sharing
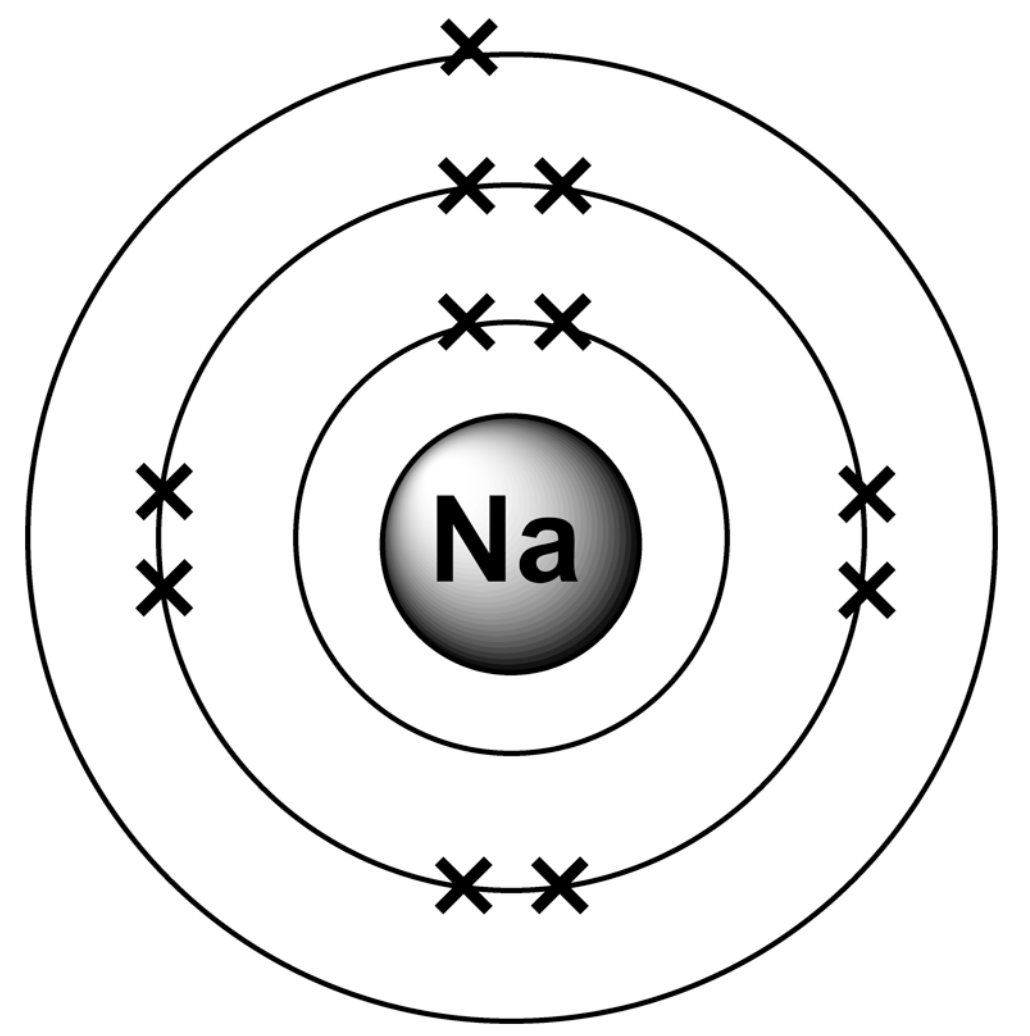
What is the electronic structure for a sodium atom?
2,8,1
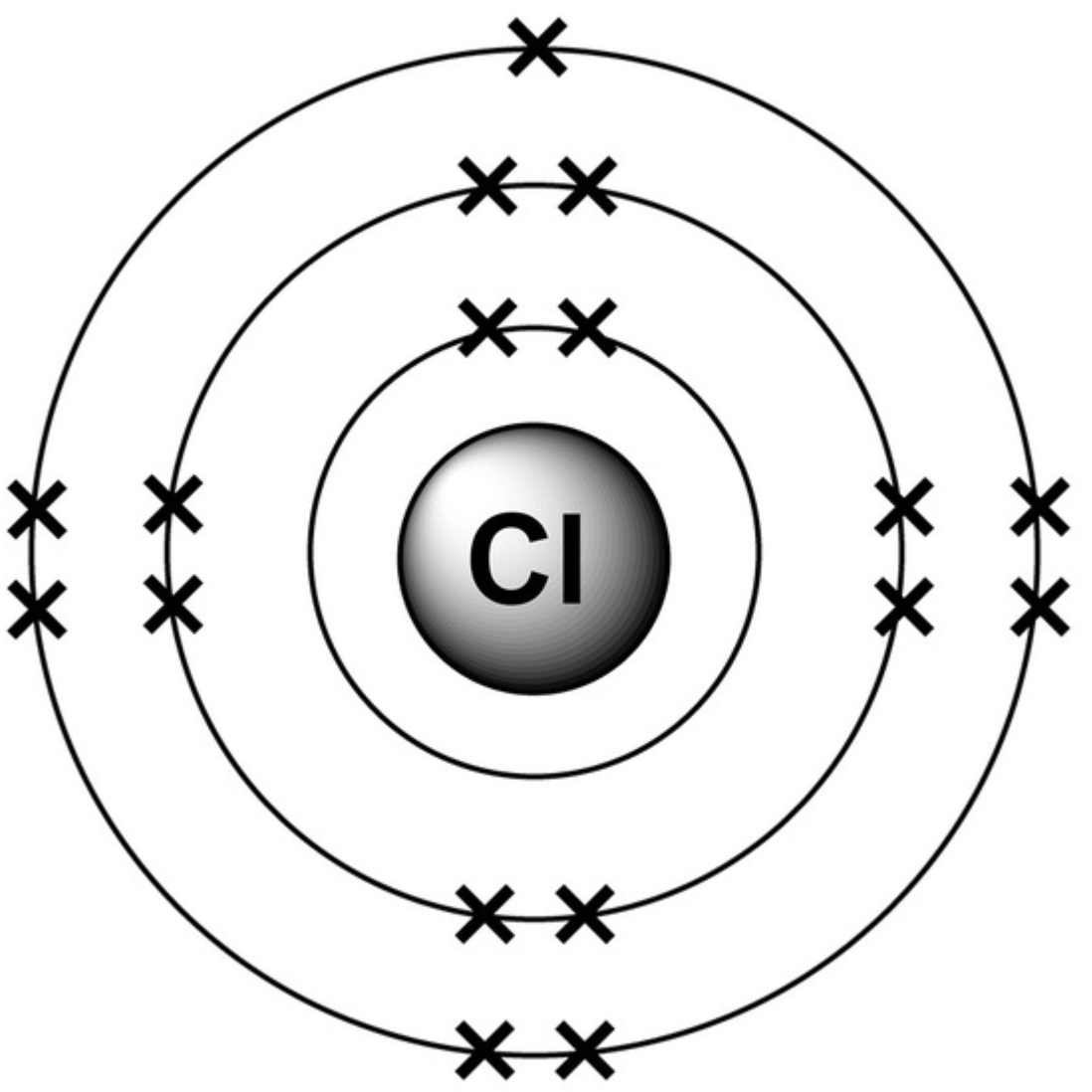
What is the electronic structure for chlorine atom?
2,8,7
- Which group and period is sodium in?
- Electron configuration = 2,8,1
- How can you tell his from the electronic structure?
- Group 1 - Since it has one electron in the outer shell
- Period 3 - since it has 3 electron shells
- Which group and period is chlorine in?
- Electron configuration = 2,8,7
- How can you tell this from the electronic structure?
- Group 7 - Since it has seven electrons in the outer shell
- Period 3 - since it has 3 electron shells
- Which group and period is oxygen in?
- Electron configuration = 2,6
- How can you tell this from the electronic structure?
- Group 6 - Since it has six electrons in the outer shell
- Period 2 - since it has 2 electron shells
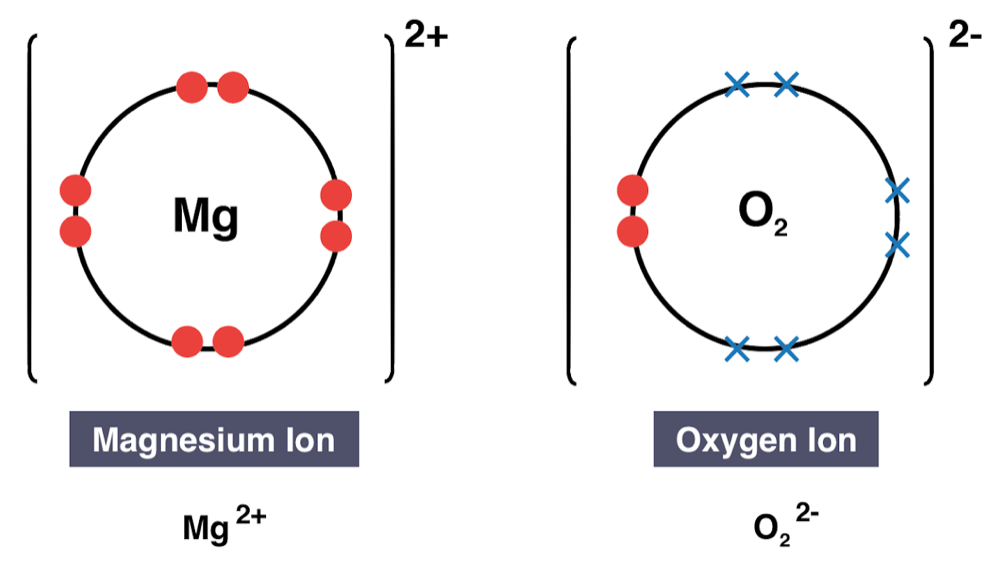
- Do metals lose or gain electrons in ionic bonding?
- Are metallic ions positively or negatively charged?
- Lose
- Positive
- Do non-metals lose or gain electrons in ionic bonding
- Are non-metallic ions positively or negatively charged?
- Gain
- Negative
- When metals from Groups 1 or 2 react with non-metals from Groups 6 and 7, which group do they then have the electronic structure of?
Noble gases (Group 0)
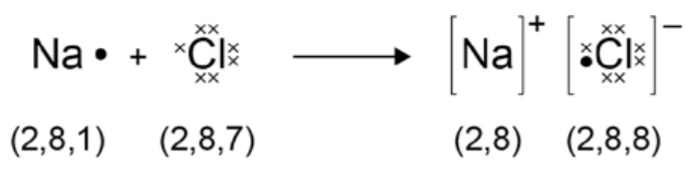
Draw a dot and cross diagram to show the electron transfer during the formation of an ionic compound of sodium chloride
Draw a dot and cross diagram to show an ionic compound of magnesium oxide
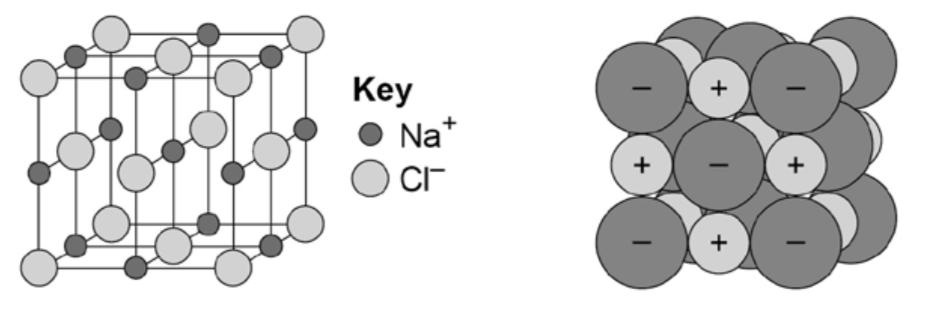
Describe the structure of a compound of sodium chloride (NaCl)
- Ionic Bonding Giant structure of ions.
- Held together by strong electrostatic forces of attraction between oppositely charged ions.
- These forces act in all directions in the lattice
Describe the giant covalent structure of a diamond
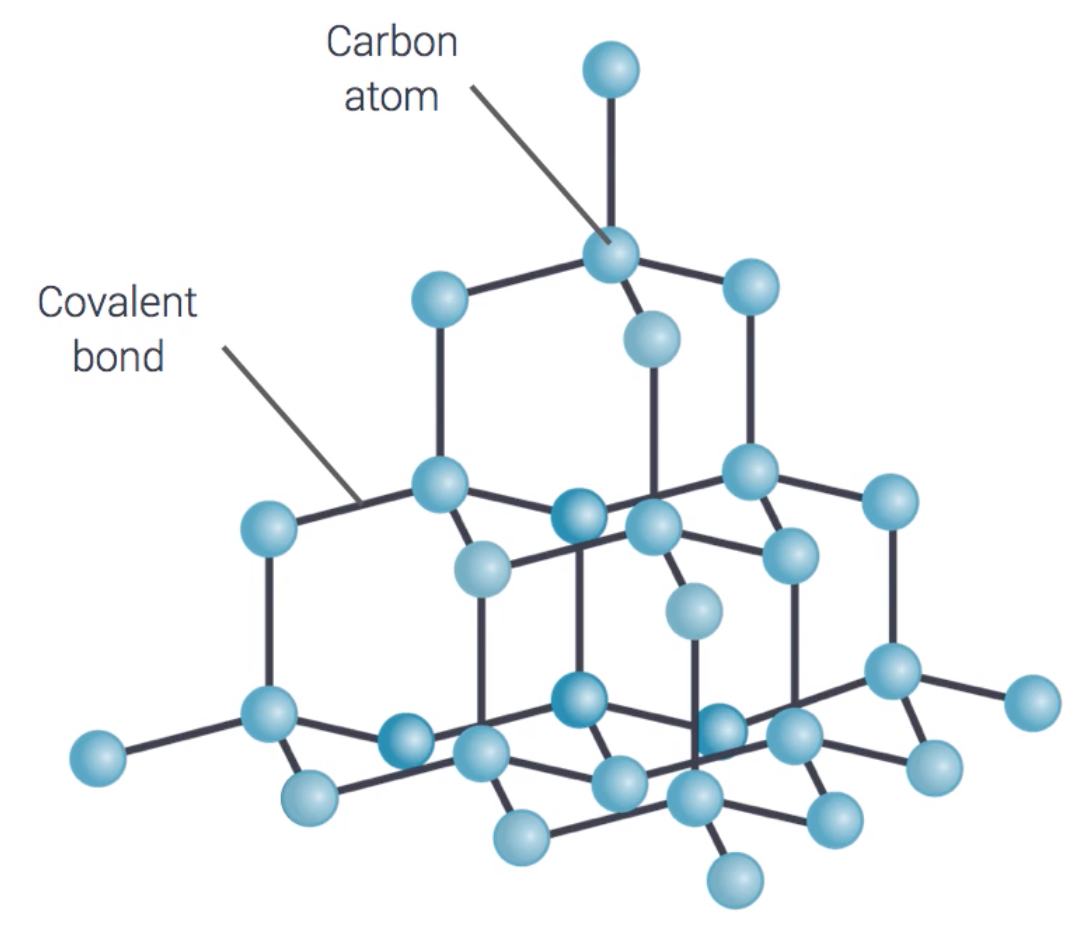
- Very strong Covalent bonds
- Only Carbon atoms
- Each carbon atom is joined to 4 other carbons
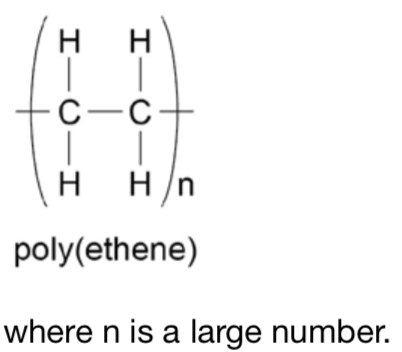
How can polymers be represented, for example a polymer of poly (ethene)
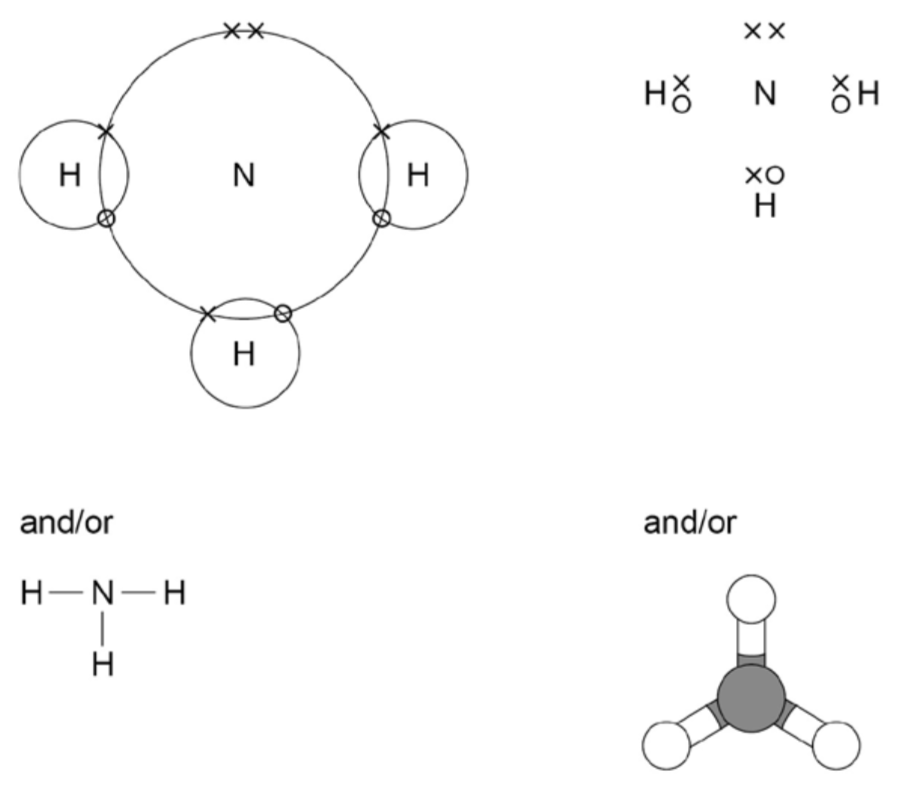
1) Draw a dot and cross driagram to represent covalent bonding in ammonia
2) How else can the bonding in ammonia be shown?