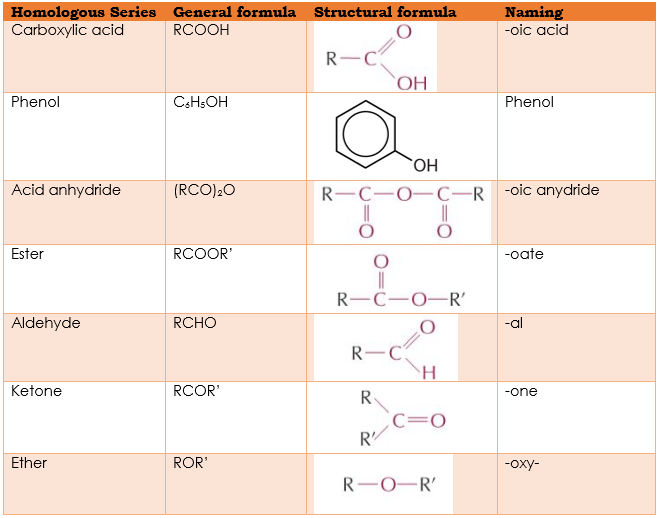
(a) the formulae of the following homologous series: carboxylic acids, phenols, acid anhydrides, esters, aldehydes, ketones, ethers
- R in general formulae represents an alkyl group (alkane missing 1 hydrogen atom)
- Each extra group (methyl/halogen etc.) on phenol means 1 less hydrogen atom on ring
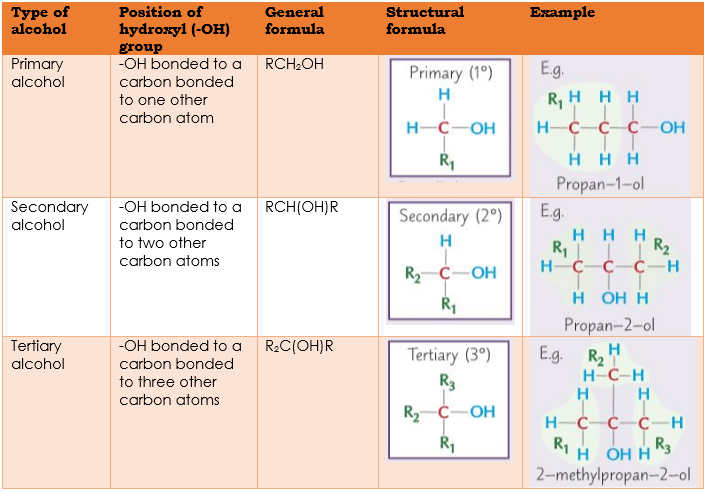
(b) primary, secondary and tertiary alcohols in terms of the differences in structures
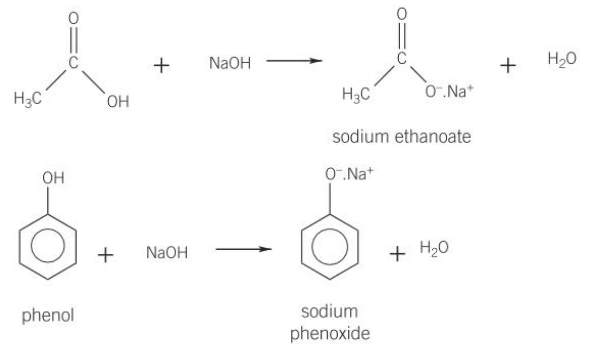
(c) (i) the following properties of phenols:
acidic nature, and their reaction with alkalis but not carbonates (whereas carboxylic acids react with alkalis and carbonates)
OH groups dissociate in water
- R-OH + H2O ⇌ R-O- + H3O+
Water dissociates to a very small extent and therefore behaves like an acid
- H-OH + H2O ⇌ H-O- + H3O+
Phenol –> equilibrium lies further to the right so more acidic than water but not as acidic as carboxylic acid
- Both phenols and carboxylic acids are strong enough acids to react with strong bases (e.g. NaOH) to form ionic salts (alcohols don’t)
- Only carboxylic acids produce CO2 (fizzing) when reacting with carbonates (alcohols + phenols don’t)
- CO32-(aq) + 2H+(aq) –> CO2(g) + H2O(l)
(c) (ii) the following properties of phenols:
test with neutral iron(III) chloride solution, to give a purple colouration
- Test for phenols:
- Mix solution with neutral iron(III) chloride (yellow solution)
- Shake
- If phenol present; purple complex forms
(c) (iii) the following properties of phenols:
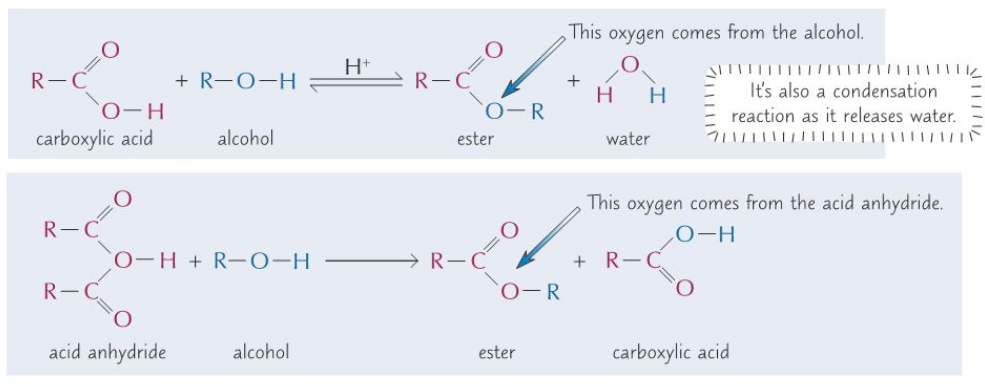
reaction with acid anhydrides (but not carboxylic acids) to form esters
- Phenols can form esters by reacting with acid anhydrides but not carboxylic acids
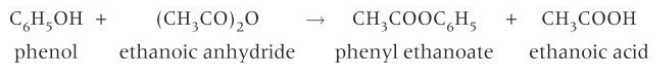
CONDITIONS + REAGENTS FOR ACID ANHYDRIDE + ALCOHOL
(d) (i) the following reactions of alcohols and two-step syntheses involving these reactions and other organic reactions in the specification:
with carboxylic acids, in the presence of concentrated sulfuric acid or concentrated hydrochloric acid (or with acid anhydrides) to form esters
- Carboxylic acid(aq) + Alcohol(aq) –> Ester(aq) + Water(l)
- Esterification reaction
- Conditions + reagents:
- Acid catalyst: conc sulfuric acid/conc hydrochloric acid
- Heat
- Acid anydride(aq) + Alcohol(aq) –> Ester(aq) + Water(l)
- Esterification reaction
- Conditions + reagents:
*
(d) (ii) the following reactions of alcohols and two-step syntheses involving these reactions and other organic reactions in the specification:
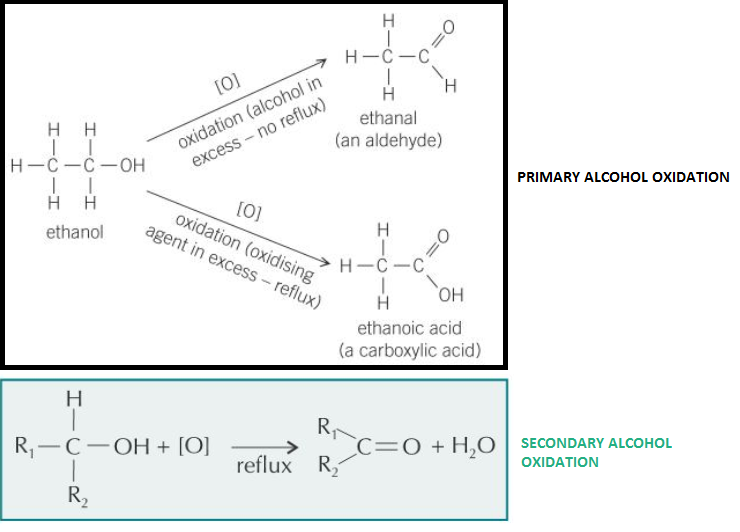
oxidation to carbonyl compounds (aldehydes and ketones) and carboxylic acids with acidified dichromate(VI) solution, including the importance of the condition (reflux or distillation) under which it is done
Primary alcohols:
- Oxidises to aldehyde then to carboxylic acid
- Orange dichromate(VI) reduced to green chromium(III) ion, Cr3+ (so reaction mixture = green)
- Conditions and reagents
- acidified potassium dichromate(VI) - K2Cr2O7
- sulfuric acid - H2SO4
- heat then distill
Secondary alcohols:
- Oxidises to ketone
- Orange dichromate(VI) reduced to green chromium(III) ion, Cr3+ (so reaction mixture = green)
- Conditions and reagents
- acidified potassium dichromate(VI) - K2Cr2O7
- sulfuric acid - H2SO4
- reflux
Tertiary alcohols:
- Don’t oxidise
- Orange dichromate (VI) not reduced (so reaction mixture stays orange)
(d) (iii) the following reactions of alcohols and two-step syntheses involving these reactions and other organic reactions in the specification:
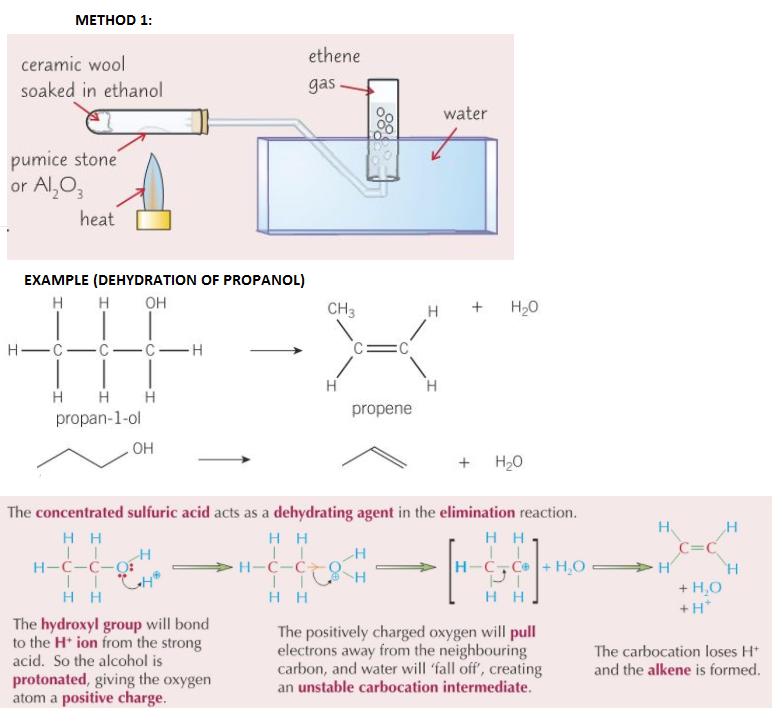
dehydration to form alkenes using heated Al2O3 or refluxing with concentrated H2SO4
- Dehydration reaction = type of elimination reaction
- 2 methods of dehydrating alkenes:
- Ethanol vapour passed over hot (300oC) catalyst (pumice stone or aluminium oxide, Al2O3) - catalyst provides large S.A for reaction
- reflux ethanol with concentrated sulfuric acid at 170oC
(d) (iv) the following reactions of alcohols and two-step syntheses involving these reactions and other organic reactions in the specification:
substitution reactions to make haloalkanes
(e) techniques and procedures for making a solid organic product and for purifying it using filtration under reduced pressure and recrystallisation (including choice of solvent and how impurities are removed); techniques and procedures for melting point determination and thin layer chromatography
- Synthesis of organic product and purification (e.g. aspirin):
Recrystallisation:
- Recrystallisation = for purifying an organic solid
- Choose a suitable solvent that this desired product dissolves in when hot and is insoluble when cold
- Dissolve the product in a minimum volume (gives a better yield) of hot solvent, so the solution is saturated
- Filter the solution to remove any insoluble impurities
- Leave the filtrate to cool until crystals form
- the impurities will stay in the solution and desired product is now solid crystals
- Remove the crystals by filtration
- Wash with ice-cold solvent and dry crystals
Melting point determination:
- Thin layer chromatography:
In thin layer chromatography (TLC), the stationary phase is a thin layer of silica (silicon dioxide) or alumina (aluminium oxide) fixed to a glass or metal plate
Draw a thin baseline in pencil 1cm from the bottom of thin layer chromatography plate
pencil is used because it will not run into the solvent
Spot a small drop of your test sample on the baseline as well as reference samples separated on the line
Place the plate in a beaker containing a small volume of solvent (this is the mobile phase) making sure the plate is suspended and does not touch the bottom of the beaker
the solvent level must be below the baseline
Cover the beaker with a watch glass
to prevent the solvent from evaporating
Leave the beaker until the solvent front is near the top
As it moves up the plate, the solvent will carry the substances in the mixture with it - but because of different affinities some chemicals will be carried faster than others and so travel further up the plate
Then remove the plate from the beaker and mark how far the solvent front has travelled up the plate before it evaporates and allow to dry
The chromatogram produced will not show the organic compounds as they are all colourless and therefore spots will need to be located using iodine, ninhydrin, or using an ultraviolet lamp
You can use the positions of the chemicals on the chromatogram to identify what the chemicals are by matching the heights reached, or Rf values with those of known compounds
(f) techniques and procedures for preparing and purifying a liquid organic product including the use of a separating funnel and of Quickfit or reduced scale apparatus for distillation and heating under reflux
(i) (i) interpretation and prediction of mass spectra:
the M+ peak and the molecular mass
- M+ peak = second peak from the right
- The mass/charge (m/z) of the M+ peak is the molecular mass
- Mass spectroscopy detects ions not molecules - so ion detected at M+ is the molecular ion, M+(g)
(i) (iii) interpretation and prediction of mass spectra:
the M+1 peak being caused by the presence of 13C
Calculations based on M+1 peak will not be required.
- First from the right peak
- Small in height in comparison to M+ peak
- Caused by presence of 13C incorporated in some of the molecular ions
(i) (ii) interpretation and prediction of mass spectra:
that other peaks are due to positive ions from fragments
- Molecule fragments into smaller ions that mass spectrometer detects
- Way in which the parent ion fragments (fragmentation pattern) is characteristic of the compound
- Base peak is the most abundant ion (giving highest intensity)
- set to 0
- other peaks are a percentage with comparison to base peak
(j) the effect of specific frequencies of infrared radiation making specific bonds in organic molecules vibrate (more); interpretation and prediction of infrared spectra for organic compounds, in terms of the functional group(s) present.
IR absorptions will be given on the Data Sheet.
- in infrared spectroscopy - a beam of IR radiation is passed through a sample of a chemical
- the IR radiation is absorbed by covalent bonds in the molecules, increasig their vibrational energy
- bonds between different atoms absorb different frequencies of IR radiation. Bonds in different places in a molecule absorb different frequencies too
- so O-H in an alcohol and O-H in a carboxylic acid absorb different frequencies
- an infrared spectrometer produces a spectrum that shows you what frequencies of radiation the molecules absorbing
- you can use it to identify the functional groups in a molecule
- also means you can tell if a functional group has changed during a reaction
- e.g. if you oxidise an alcohol to an aldehyde, O-H absorption would disappear from spectrum and C=O would appear
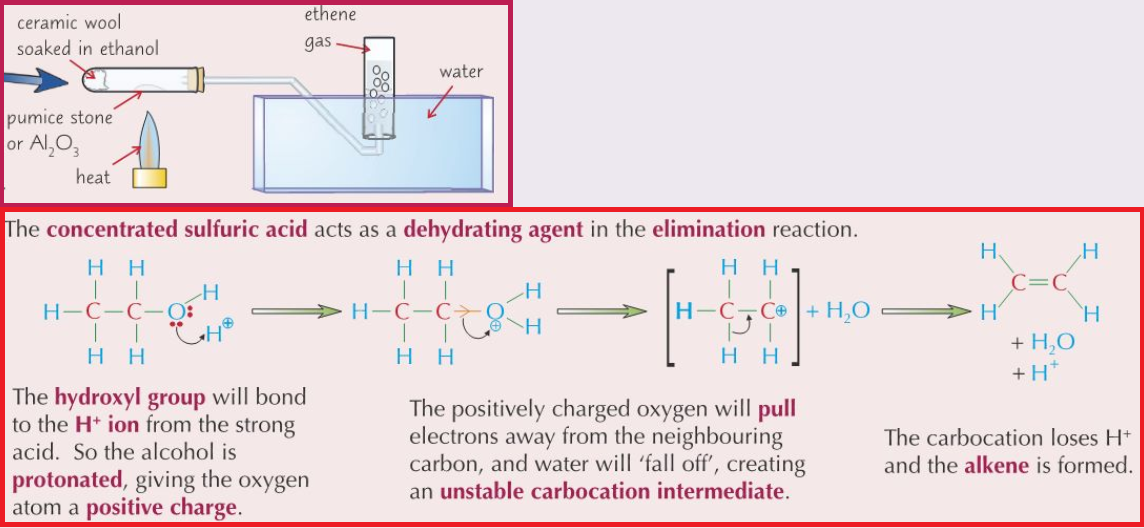
(h) the term elimination reaction
Example: alkenes from alcohols.
- Elimination reaction: A reaction where a small molecule is removed from a larger molecule and not replaced.
- Reverse of addition reaction
- e.g. Dehydration (removal of water) of alchols (going from alcohol to alkene)
- C2H5OH –> CH2CH2 + H2O
- Reverse of addition reaction
- Conditions and Reagents for dehydration of alcohol
- Either reflux alcohol vapour with concentrated sulfuric acid (170oC)
- or Alcohol vapour passed over a hot (300oC) catalyst (pumice stone/aluminium oxide, Al2O3)
- catalyst provides a large surface area for reaction
(g) the principles of green chemistry in industrial processes
Learners should be able to make suggestions based on (but not to quote verbatim) the 12 ‘principles of green chemistry’. Learners will be expected to analyse and use given information.
- Green chemistry is about
- being more sustainable (using renewable resources, like bioethanol instead of fossil fuels, or solar power)
- reducing waste produced (less steps, better atom economy)
- lowering cost
- using less energy (catalysts)
- reducing impact on the environment (biodegradable products)
- and health and safety (safer chemicals + reactions)


