Theme 1: Extreme Environments Flashcards
Commercial aircraft are pressurised to a pressure that is equivalent to an altitude of…
5000-8000ft
What is the atmospheric pressure at sea level (in mmHg)?
760mmHg
At what altitude (in feet) is the atmospheric pressure half of that seen at sea level?
18,000ft
What equation can be used to understand all of the physiological changes at altitude?
[EXTRA]
- The alveolar gas equation
- You can understand the changes by remembering that Patm decreases, PH20 stays the same and PaCO2 increases slightly.
- This means that the PAO2 greatly falls and therefore haemoglobin saturation falls too. CO and ventilation can be increased to compensate.
- Ventilation decreases PaCO2, which helps.

Why are aircraft pressurised to a pressure equivalent to 5,000-8,000ft?
- This leads to a partial pressure of oxygen in the arterial blood that corresponds to a high haemoglobin saturation.
- At higher altitudes, the haemoglobin saturation rapidly falls off.

What is Boyle’s law and how is it relevant?
It relates to travel to high altitudes and can explain certain pathologies of altitude.

Give an example of a disease that can be exacerbated by travel to altitude.
- Tension pneumothorax
- Since gases expand at altitude, a tension pneumothorax can become even more dangerous at altitude
Does hypoxic pulmonary vasoconstriction occur during air travel?
First there was no certain evidence:
- (Luks, 2009) suggests that patients with a mean pulmonary arterial pressure of >35mmHg should avoid travel to more than 2000m altitude.
- However, much of this understanding was based off of studies using cat lungs -> There are significant differences between species so we do not know is HPV begins in an aircraft cabin.
- Case reports suggest that there may be a problem -> (Toff, 1993) describes an obese woman and (Noble, 1999) describes an individual with kyphoscoliosis who both had problems at altitude
Then there were studies carried out:
- (Smith, 2012) studied a healthy patient in flight between London and Denver, as well as during the stay in Denver. They showed increased systolic pulmonary artery pressure in-flight and in Denver. But none of this was above the threshold for pulmonary hypertension (36mmHg).
- (Smith, 2013) carried out a similar experiment in a patient with Chuvash polycythaemia on a flight between London and Dubai. They showed greatly increased systolic pulmonary artery pressure in-flight, far above the threshold for pulmonary hypertension (36mmHg).
- This suggests that certain patients may be more susceptible to HPV than others.

Give some evidence for high altitude pulmonary oedema (HAPE).
(Gabry, 2003):
- Studied subjects ascending the French Alps
- They showed evidence of pulmonary oedema even below 2,400m
How does HPV compare in young and older patients? When is this relevant?
(Turner, 2015):
- Used a hypobaric chamber to simulate flight
- Compared arterial oxygen saturation and systolic pulmonary arterial pressure between patients in the 19-24 and 61-69 age groups
- The older group had a greater increase in pulmonary arterial pressure, but also lower oxygen saturation, so it was unclear if older patients show more HPV or whether this was simply due to greater hypoxia
(Balanos, 2015):
- Clarified this uncertainty by controlling for hypoxia between the groups
- Found that older patients do indeed have greater HPV

What are some physiological factors that affect the human nody during space flight?
- Ionizing radiation
- Microgravity
- Vacuum of space
- Extreme temperature
What are some sources of ionizing radiation that the human body may be exposed to during space flight?
- Solar wind (lower energy) -> No risk
- Solar particle events (intermittent, high-energy) -> Risk of acute radiation syndrome
- Galactic cosmic radiation (very high energy) -> Increased risk of cancer
What are some effects of microgravity on the body?
- Vestibular (balance) system
- Fluid redistribution
- Deconditioning -> Bone, Muscle, Cardiovascular system
- Visual impairment
- Other effects (anaemia, immune dysfunction, back pain, sleep disturbance, etc)
What is space adaptation syndrome?
- “Space sickness”
- It is the sickness that 50-70% of astronauts experience upon space travel, lasting around 2-3 days
At what rate does bone loss happen in space and what are the consequences?
- About 1% per month in weight-bearing bones
- There is increased risk of fractures and kidney stones
What are some active countermeasures against musculoskeletal and cardiovascular deconditioning?
- Treadmill and cycle exercise devices for aerobic fitness and postural muscles
- Advanced Resistive Exercise Device (ARED) for muscle and bone
The cost of these is time, oxygen, food and water.
Describe the visual impairment that astronauts often develop.
- Space flight-associated neuro-ocular syndrome (SANS)
- Previously called visual impairment and intracranial pressure syndrome (VIIP)
- Approximately 50% of ISS astronauts report impairment of vision
- Some have eye abnormalities such as swelling of the optic disc and flattening of the eyeball
- Uncertain aetiology
What are some problems that astronauts may experience upon return to gravity?
- ‘Entry adaptation syndrome’ (like space sickness)
- Balance problems
- Orthostatic intolerance
- Reduced aerobic capacity
- Need for rehabilitation
What are some ways of simulating microgravity?
- Bed rest with 6˚ head-down tilt (‘pillownauts’)
- Parabolic aircraft flights
- Water immersion (neutral buoyancy)
What are some signs of cephalic body fluid shift in microgravity?
- Fullness in the head
- Nasal congestion
- Objective facial oedema and erythema
- Volume of lower extremities begins to diminish
- Superficial vascular system of upper body seen to engorge
- Feeling of facial fullness causing discomfort
- Jugular venous distension along entire length of neck
- Increase in jugular vein cross section via ultrasound
How does CVP change in space?
- It would be expected to increase since stroke volume and CO increase
- BUT (Buckey, 1996) used a central venous catheter to study CVP and in fact found that it increases
- This is suggested to be due to decreased tissue compression
Give some experimental evidence for the G-forces a person might experience during a suborbital flight. Why is this relevant?
(Blue, 2014):
- Used a centrifuge to evaluate how potential commercial space flight participants might be affected during launch, etc. and how this would affect their medical conditions
- The forces are more Gx (front-to-back direction) rather than Gz (head-to-toe direction), unlike a fighter plane pilot may experience (since space flight involves being reclined)
- The front-to-back direction of the forces can lead to chest compression and thus difficulty breathing

What is an example of an organ that may be affected by high G forces?
Lungs
Name some studies that investigated the effects of spaceflight on various physiological parameters and anxiety.
- (Blue, 2012) -> Studied arterial oxygen saturation
- (Blue, 2014) -> Studied arterial oxygen saturation in participants with various medical conditions
- (Blue, 2017) -> Studied anxiety and task performance
- (Pollock, 2021) -> Studied a wide range of parameters (GOOD STUDY)
Describe an experiment about the effects of spaceflight on arterial oxygen saturation.
(Blue, 2012):
- Used a centrifuge to simulate spaceflight, at various Gz (head-foot) and Gx (front-back) G-force values
- Studied 77 participants, aged 22-88
- 16 participants were studied using pulse oximetry
- No reading below 89% was obtained during any centrifuge exposure
- 8 had desaturation to 92% or below at least once (mostly related to peak +Gx)
- 5 had repeat desaturations
- +Gz did not result in significant changes in pulse oximetry
- In summary, of those studied, half experienced desaturation to 89-92%
(Blue, 2014):
- Carried out a similar experiment in particular in participants with medical conditions (e.g. hypertension)
- Combined +Gx and +Gz, with a peak of +6.0 Gx/+4.0 Gz
- 69% got greyout
- 6% got chest discomfort
- Minimum arterial oxygen saturation was recorded was 90% and no clinical symptoms of hypoxia were noted at any time
The studies established the difficulty of finger probe pulse oximetry was of limited utility due to difficulty in keeping the probe on. Also, there were few low SpO2 readings regardless of G magnitude or direction, so it is unlikely that SpO2 monitoring during commercial spaceflight will be of any greater utility.’
Describe an experiment about the effects of spaceflight on anxiety and task performance.
(Blue, 2017):
- Used a centrifuge to simulate spaceflight
- After the centrifuge stopped, auditory alarms sounded and subjects were verbally instructed to complete a series of tasks (total of 12 steps), including removing their harness, reorienting gondola air vents, signalling one of the gondola cameras, and replacing their harnesses.
- Heart rate increased by 34%
- On average, subjects made 3 mistakes
- Only 28/137 (20%) made no mistakes during the scenario
Describe an experiment that studied the effects of spaceflight on a number of physiological parameters.
(Pollock, 2021):
- Used a centrifuge to stimulate spaceflight at Gx (front-back) values of +2, +4 and +6
- Performed all studies with normal air and also with 15% oxygen to simulate the way in which the cabin could be pressurised.
- Arterial oxygen saturation fell at +4 and +6 Gx values. The saturation was overall lower when breathing 15% oxygen.
- The arterial partial pressure of oxygen decreased and the alveolar-arterial gradient increased with increasing Gx force. These are evidence of impaired V/Q matching.
- Ventilation was relatively unaffected until +6 Gx, when it began to increase slightly. This was because, although respiratory rate increased with Gx, the tidal volume decreased.
- The neural respiratory drive increased greatly with Gx, as did the work of breathing.
These results point to the idea that the Gx force compresses the lungs, impairing function. Thus, respiratory drive increases but there is persistent feeling of breathlessness. This could partly explain the anxiety that many participants experience. The respiratory problems are exacerbated by airline-style cabin pressurisation.

Describe an experiment that studies the effects of spaceflight on ventilation in different parts of the lungs.
(Pollock, 2021):
- Simulated spaceflight using a centrifuge
- Divided the chest into 8 sections
- As Gx (front-back) G force increased, the ventilation became more ventral
- However, there was also more residual air left in the ventral areas of the lungs at the end of expiration

Respiratory disruptions in response to Gx forces are somewhat analogous to…
Temporary COPD and obesity (transient respiratory failure)
How many compartments do we consider the body to have in temperature regulation?
- Two compartment system:
- The core (brain, thoracic and abdominal compartments containing the heat producing organs)
- This region is held at ~37.0+/- 0.5*C
- The peripheral shell (this is cooler and allows for regulation of temperature)
- The core (brain, thoracic and abdominal compartments containing the heat producing organs)
- Communication between the two compartments is achieved via the circulation as blood is allowed to move between the two compartments, and can do so in either direction
What is a homeotherm?
- This is an organism that maintains a constant body temperature through metabolic processes
- This body temperature is often above that of the surrounding temperature
- Humans are homeotherms
What is the thermoneutral zone?
- This is the temperature range over which there is no ‘active’ temperature gain or loss
- This means that the surrounding temperature is pretty well matched with our typical heat generation
- If temperature drops below this zone, we start to increase heat production to maintain homeostasis
- If the temperature increases above the thermoneutral zone, we begin to actively dissipate heat and begin cooler temperature-seeking behaviours
What is the thermoneutral zone for humans?
- Naked = 25-30*C
- Light clothing = 18-22*C
- Affects heat loss from the body therefore reduces the range
What hormones affect BMR?
- Thyroid hormone
- Testosterone
- Growth hormone
How does thyroid hormone (thyroxine) affect BMR?
- Thyroid hormones (thyroxine) can raise BMR to 50-100% above normal
- Total loss of thyroxine will decrease BMR to 40-60% of normal levels
[EXTRA]
- Adaptation of the thyroid gland is seen in people living in different geographical locations
- People living in arctic regions have a BMR 10-20% above those in tropical regions, allowing an increased generation of heat to deal with the colder environment
How does testosterone affect BMR?
~10-50% increase in BMR
How does growth hormone affect BMR?
~15-20% increase in BMR
How does the content of the meal affect changes in BMR?
- Fat/carbohydrate meals cause BMR to increase by ~4%
- Protein heavy meals can raise BMR up to 30% (hence the meat sweats due to raised body temp after raised BMR)
What is core body temperature?
Between 36.1-37.8*C in normal physiological parameters
NB that this can differ from the peripheral temperature, which is (understandably) cooler
At what temperatures can a naked person in dry air maintain a ‘normal’ core body temperature?
Between approx. 12*C and 54*C (this is larger than the range for the thermoneutral zone as heat loss and gain processes are considered)
What are some normal variations in body temperature?
- Circadian rhythms
- Daily variations in body temperature occur, +/-0.5*C
- Minimum occurs in the early hours of the morning, peak in the late afternoon
- Menstrual cycle
- Rise of +1*C is seen following ovulation

Approximately how much of daily energy expenditure is accounted for by basal metabolism?
~60%

What is Kleiber’s law?
[EXTRA]
- ‘For the vast majority of animals, an animal’s metabolic rate scales to the 3/4 power of the animal’s mass’
- I.e. if q0 is the animal’s metabolic rate, and M the mass, then Kleiber’s law states that q0 =~M3/4
- Essentially, larger animal = larger metabolic rate
- Named after Max Kleiber, Swiss agricultural biologist
- There is still some argument over the exact value of the power, with some claiming it to be 2/3, but the constant is relatively arbitrary

What is the largest variable for heat production?
Physical exercise
- ~7% of daily energy expenditure is due to ‘non-exercise’ activity (e.g. fidgeting, non-specific exercise, muscle tone required for holding posture)
- As exercise increases/becomes more significant, there is a rapid increase in energy expenditure requirements

What is brown adipose tissue (BAT)? How does it generate heat?
- Adipose tissue rich in mitochondria
- Also contain the uncoupling protein 1 (UCP-1)
- Heat is generated through the dissipation of the proton gradient across the inner mitochondrial membrane via UCP-1
- Role in adults is a topic of ongoing research (particularly in isolating treatments for obesity) but it has been shown to play a vital role in neonatal thermogenesis (as neonates are unable to shiver)
- Found primarily between the shoulder blades and around the kidneys

When is brown adipose tissue (BAT) activated? Through what mechanisms is this achieved?
- BAT is activated in resposne to low temperatures
- Stimulation occurs via the release of noradrenaline from the sympathetic nervous system
- Activation acts via multiple transcription factors, including PGC1alpha and the PPARs to increase mitochondrial proliferation and fatty acid metabolism
- PPARs = peroxisome proliferation activated receptor

What are the 4 heat loss mechanisms?

What law is used to describe heat losses due to radiation?
Stefan-Boltzmann law
- Emissivity is the measure of a material’s effective ability to emit and absorb thermal radiation, black objects have a high emissivity, white objects do not
-
NB That the temperature difference between the object and the environment is to the power of 4, therefore has a large effect on the equation
- Also note that energy can only be lost from the body via radiation if the temperature gradient favours energy loss to the surroundings (i.e. they are cooler than the body)
- If the surrounding temperature increases to exceed that of the body, radiation will occur in the other direction/heat is transferred to the body

What are arterio-venous shunts?
- These are methods of allowing a separation between nutritional and thermoregulatory blood flow
- These vessels bypass the capillaries and can increase or decrease blood flow to the periphery to facilitate or limit heat loss respectively
- Flow through shunts can be ‘on’ or ‘off’
- Blood flow through the shunts can be 100-fold greater than nutritional requirements
- Shunts are controlled by centrally mediated alpha-1 adrenergic receptors
- Although this is augmented by local alpha-2 adrenergic receptors in the periphery

How are changes in temperature sensed?
- Primarily through nervous feedback systems from the periphery to the CNS
- Centre in the brain: temperature regulating centers in the hypothalamus
What is the key centre for temperature regulation in the brain?
Hypothalamus is area through which almost all temperature regulating mechanisms work
What is the ‘thermostat’ for the body?
- The preoptic and anterior hypothalamus operates as the body’s thermostat
- This part of the brain has greater sensitivity to heat than to cold
Where are the peripheral temperature sensors located and what are they more sensitive to?
- Spinal cord, abdominal viscera and great veins (deep body sensors)
- Skin
- Greater sensitivity to cold than hot
Are the following more sensitive to hot or cold stimuli?
- Preoptic and Anterior Hypothalamus
- Peripheral temperature receptors
- Hot
- Cold
How do peripheral temperature receptors respond to changes in temperature?
- They alter their discharge frequency
- Sensors are rapidly adapting/will rapidly adapt to new static levels
- This means that they are bad at sensing absolute temperature, but are very good at sensing changes in temperature
What is a simple test to show the adaptive nature of peripheral temperature receptors?
[EXTRA]
- Two bowls of water, one quite warm (35-40*C), the other very cold (add ice, ~0*C)
- Put a hand in each boel and leave it there for 3-4 mins, then put the hand into a bowl of water at room temperature
- Hand in ice water thinks that the bowl of room temperature water is really hot, hand in bowl of warmer water thinks that room temperature bowl is very cold
- This shows that the change in temperature has shaped the response, not the actual temperature, or else the hands would experience the same sensation
What is the function of the posterior hypothalamus in relation to temperature regulation?
- This part of the brain collates and combines the signals from the peripheral sensors and from the central sensors (in the anterior hypothalamus)
- This leads to control over the heat-conserving and heat-producing reactions of the body
How much can heat production be increased by through shivering?
~200%
When can shivering not occur?
In newborn infants - this makes them far more susceptible to the cold and they instead rely upon the action of BAT
What is shivering?
- Rapid (~50Hz) and unsynchronised muscular activity
- This results in ATP cross-bridge cycling to facilitate contraction and production of heat as a byproduct of the metabolic reaction
- Shivering is likely to result from a feedback oscillation of the muscle spindle stretch reflex mechanism
- Random oscillation is overlaid on a centrally controlled 4-8 cycle/min of ‘waxing and waning’
- E.g. shivering cycles between more and less severe bouts
Where is the need for shivering signalled from? Where is this signal translated?
- Posterior hypothalamus signals the need for shivering after collating inputs from peripheral receptors and central receptors (anterior and preoptic hypothalamus)
- This signal is translated to the anterior motor neurons
What are the differences in sweat composition when comparing light and heavy stimulation of sweat secretion?
- With light stimulation, almost all of the Na+ and Cl- is reabsorbed (along with a lot of water) as the secretions travel along the duct
- This leads to a concentrated solution made up of urea, lactic acid and potassium
- With heavy stimulation, large volumes of sweat are produced
- This results in poor reabsorption of NaCl and increased fluid loss (due to the subsequently less concentrated liquid secreted)
What is the effect of hot climates on sweat production?
- Over 1-6 weeks of exposure to a hot climate, sweat production is upregulated
- In response to the increased loss of NaCl caused by this change, the body also upregulates aldosterone secretion to increased Na+ reabsorption
What is hypothermia?
- This is where continued exposure to cold temperatures causes the core body temperature to drop below 35*C
- Energy expenditure falls 13% for every 1*C drop in core temperature
- Loss of consciousness occurs at core body temp of 30*C
What are the cardiac changes seen in hypothermia?
- Changes can be seen on the ECG, including bradycardia and prolongation of the PR and QT intervals
- Ventricular fibrillation occurs at 28*C
Which groups of people are most susceptible to hypothermia?
[EXTRA]
- Neonates are particularly susceptible due to reduced behavioural adaptations possible (e.g. cannot put on clothes or move) and have a greater SA:Vol ratio than adults
- They also cannot shiver, but do have BAT for thermogenesis
- The elderly are also susceptible due to potentially reduced mobility and less efficient thermoregulatory mechanisms
- Lessened mobility also makes them less able to move for heat generation and also slows the process to find and put on clothes
What is the effect of infection on temperature regulation?
- Infection can lead to an elevation of the core body temperature set point, and therefore hyperthermia
- This is caused either through the direct action of proteins/ protein breakdown products/lipopolysaccharide toxins released from bacterial cells (pyrogens) or by their action on macrophages, leukocytes and lymphocytes
- This causes the release of cytokines (IL-1 and IL-6)
- These pyrogens act to elevate prostaglandin E2 synthesis in the hypothalamus (this raises the set point)
What molecule causes the set point of body temperature to be raised in the hypothalamus?
- Prostaglandin E2 (PGE2)
- Acts on E2 receptors in the anterior hypothalamus to raise the set point of body temperature
What are antipyretics?
- Drugs that act to reduce the elevated set point
- Examples include:
- Acetaminophen (paracetamol)
- Aspirin and other NSAIDs
How are antipyretics thought to work?
- Principally through their inhibition of the enzyme cyclooxygenase (COX), preventing the synthesis of inflammatory prostaglandins (including PGE2)
- Recent studies on the mechanism of action have revealed effects independent of COX inhibition as well
What are the effects of continued exposure to high environmental temperatures?
[EXTRA]
- Heat exhaustion
- Heat stroke
What is heat exhaustion?
- AKA heat syncope
- Occurs due to hypovolaemia following excessive sweating
- Symptoms include hyperthermia, excessive sweating, dizziness, nausea, headache
What is heat stroke?
[EXTRA]
- This is where body temperature >40.6*C
- Thermoregulatory mechanisms fail at this point, causing the patient to become dizzy and confused
- Loss of consciousness follows rapidly, then death
- Treatment involves rapidly reducing body temperature
- There is some disagreement over whether cold or cool water should be used
- Cold water can induce vasoconstriction and shivering, which will instead elevate temperature
What is malignant hyperthermia (MHS)?
[EXTRA]
- Malignant hyperthermia is a pathological state where body temperature is overwhelmed by heat production
- It is often fatal
- Caused by drugs include the muscle relaxant suxamethonium and volatile anaesthetics including halothane
- This is very rare (1 case per 100,000 in adult general anaesthetics)
- Can be caused by mutations in ryanodine receptor 1 (RYR1), inherited in an AR fashion
- Dantrolene is the reversal agent for MHS caused by anaesthetic agents, and so will be found in the collection of all anaesthetists
- Some individuals with this condition can occasionally enter malignant hyperthermia after extreme exercise or heat stroke, as the condition is caused by over-activation of skeletal muscle and its contraction, causing excessive heat release
What is the effect of burn injuries on thermoregulation?
- Burns cause cell damage and a hypermetabolic state
- This is characterised by increased energy expenditure and increased muscle protein catabolism, which can lead to a hyperthermia of sorts
- If there is extensive skin loss, however, hypothermia can occur as the skin is essential for many energy- and heat-preserving behaviours
- With increased energy expenditure, more heat will be lost and so burns can rapidly lead to hypothermia
How can sweat production be increased? What properties does this secretion have?
- Sweat production can be increased by cholinergic sympathetic nerve fibres
- The glands can also be stimulated by adrenaline/noradrenaline, e.g. during exercise to increase heat dissipation
- This elicits the secretion of a primary fluid into the sweat duct
- This fluid has a composition similar to that of plasma, just without plasma proteins
- As regulation/reabsorption occurs along the tubule, if the demand for secretion increases and therefore the transit time decreases, then the time over which this secretion can be modified is rapidly decreased

What is frostbite?
- Severe form of hypothermia
- Occurs when surface areas (commonly on peripheral parts of the body) freeze and there is formation of ice crystals
- Commonly affects the hands, feet and ear lobes
- If extensive formation of ice crystals occurs, this can lead to cell death
- Therefore gangrene often follows thawing
- Surgical removal of affected areas is necessary

What happens when the set point for body temperature is raised during infection?
- Individuals feel ‘cold’ despite core body temperature being above the normal level
- This results in the recruitment of normal physiological responses to being cold, such as shivering
- Upon restoration of a normal set-point, individuals experience a febrile ‘rush’ or ‘crisis’ where the body tries to restore a normal core temperature through sweating and vasodilation
- This is seen at the end of illness, but there is also some evidence to suggest that this occurs cyclically throughout fever (bouts of breaking and shivering)

Draw a summary of thermoregulation.
- Peripheral and central receptors detect temperature
- It information is integrated in the hypothalamus, which has a set point that is dependent on multiple factors, such as pyrogens
- The hypothalamus then outputs to effecotrs via the cerebral cortex, sympathetic nerves and motor nerves

What is a good way of making someone cold (for study)?
Cold water immersion
How are non-noxious cold stimuli detected?
- TRPM8 channels in the CNS and periphery
- The identification of the molecular basis of temperature sensing could have therapeutic implications
Describe the initial physiological response to cold water immersion (cold shock). Give some experimental evidence for this.
(Keating, 1964):
- Used a cold shower to simulate cold water immersion
- PCO2 decreased -> This shows an increase in ventilation
- Arterial pressure increased -> This shows peripheral vasoconstriction
(Cooper, 1976):
- Water immersion leads to increased tidal volume and ventilation
The peripheral vasoconstriction is useful but it is less clear why there is increased ventilation. The ventilation is significant because it increases the risk of water inhalation.

What is the diving reflex?
- Facial (trigeminal) immersion triggers the ‘diving reflex’, which causes bradycardia, increase in stroke volume and peripheral vasoconstriction.
- This is not necessarily useful for temperature regulation. It is more likely a residue evolutionary reflex.
Give some experimental evidence for the diving reflex.
(Heistad, 1968):
- Measured heart rate and blood pressure in humans when holding breath and during facial immersion
- Heart rate dropped mildly when holding breath and more severely during facial immersion
- Blood pressure increased mildly when holding breath and more severely during facial immersion
- Blood flow to the fingers decreased in both cases, but blood flow to the forearm decreased only during facial immersion

Describe the idea of autonomic conflict in cold water immersion.
- During cold water immersion, the cold water reflex leads to sympathetic stimulation of the heart, while the diving response leads to parasympathetic stimulation of the heart
- These opposing activities can pre-dispose to cardiac arrhythmias.

Are early cold responses (e.g. heart rate and ventilation) fixed? Give some experimental evidence for this.
- No, habituation can happen with repeated exposure to cold water
- (Tipton, 1988) studied heart rate and ventilation in response to cold water immersion. The response decreased when repeated for several days in a row.

How does habituation of cold responses (e.g. heart rate and ventilation) happen? Give some experimental evidence for this.
- The effect seems to be mediated centrally (rather than at the level of the peripheral receptors)
- (Tipton, 1998) exposed only the left half of the body to the cold, repeatedly. This led to habituation of both sides of the body to the cold, suggesting that habituation is a central process.
How long can habituation of cold responses last?
Several months
What is meant by cross-adaptation with reference to cold responses?
(Lunt, 2010):
- Found that, for example, there may be a change in the response to hypoxia also after habituation to cold exposure
- Thus, there may be adaptation that affects multiple stresses
Is habituation of cold responses all physical?
There may be a psychological component to cold habituation, and/or some psychological benefit (van Tulleken, 2018)
Summarise the pathological effects of hypothermia.

Summarise the physiological responses to pulmonary oedema.
- Behavioural: including exercise, which may be effective at heat production, but could increase convective heat loss
- Vasoconstriction: cutaneous vasoconstriction is mediated by the sympathetic nerves, diverting blood away from the skin
- Shivering: helpful for heat generation in the short term, but costly in energy and fuel usage (and not generally favoured by populations adapted to life in cold climates)
- Non-shivering thermogenesis: heat generation by metabolic means other than muscle movement, mainly in brown adipose tissue in neonates, partly under hormonal control (thyroxine)
What is frostbite?
- When cold-induced peripheral vasoconstriction decreases blood flow to the extremities, such that there is an increased risk of cold damage.
- Some groups (e.g. fishermen) may have down-regulated peripheral vasoconstriction in order to avoid this and maintain dexterity in the cold.
What is swimming-induced pulmonary oedema?
- A rare event similar to HAPE, except caused by cold water immersion
- It may involve acute breathlessness and blood tinged sputum
- It may be due increased central blood volume (peripheral vasoconstriction) and elevated wedge pressure (ventricular dysfunction), leading to ‘stress failure’ of pulmonary capillaries.
- (Paz, 2020) provide a case report of a 58-year-old female athlete presenting with acute dyspnea during the swimming portion of a half-triathlon competition.
What tissue is responsible for adaptive thermogenesis?
Brown adipose tissue
Is brown adipose tissue found in adults?
Yes, it was previously thought to be just in neonates, but it is now thought to also play an important role in responses to cold in adults.
Describe an experiment showing the importance of brown adipose tissue in adaptive thermogenesis.
(Vertanen, 2009):
- Studies 5 adult volunteers studied with PET-CT scans using F-FDG (a radiotracer that marks the uptake of glucose)
- Carried out a warm scan and cold scan
- Prior to ‘cold’ scan, subjects’ feet were exposed to iced water intermittently for 2h, while sitting in a room at 17-19ºC
- The cold scans showed greater glucose uptake than the warm scans, particularly in the brown adipose tissue -> This is an indicator of its role in adaptive thermogenesis
- Three volunteers had biopsies of the cold-induced areas of adipose tissue, which showed high expression of UCP1 protein -> Thus UCP1 is expressed in BAT

Give an example of acclimatization to cold.
Some groups (e.g. fishermen) may have down-regulated peripheral vasoconstriction in order to avoid frostbite and maintain dexterity in the cold.
Give an example of population-level adaptation to cold. Give some experimental evidence.
(Cardona, 2014):
- Carried out a GWAS of cold adaptation in indigenous Siberian populations
- Found a number of genes on chromosome 11 that have been subject of positive selection
(Clemente, 2014):
- Studied the gene CPT1A, which is a regulator of cellular metabolism and helps to regulate brown adipose activity.
- This gene may even have undergone positive selection despite some adverse effects on energy balance.
Give an example of how adaptations to cold could be used as a therapeutic treatment for obesity.
Topical menthol could stimulate thermogenesis.

Describe the cytoprotective effects of hypothermia and why this is important.
- In 1999 the Swedish doctor Anna Bågenholm made a full recovery after being frozen for 80 min under a layer of ice (core temperature 13.7ºC), likely due to the cytoprotective effects of tissue hypothermia (Gilbert, 2000)
- This is exploited for therapeutic benefit during neurosurgical or cardiac procedures, and therapeutic cooling (e.g. 33ºC) can improve neurological outcome following cardiac arrest (Lascarrou, 2019)
At what temperatures is hyperthermia classified?
- The hyperthermic range is >40ºC, when neurological manifestations occur including confusion, agitation and slurred speech.
- Serious illness or death from ‘heatstroke’ is likely with prolonged exposures >40.5ºC.
Describe how heat acclimation happens. Give some experimental evidence.
(Robinson, 1943):
- Exposed subjects to a desert-like environment
- Over 24 days of exposure, the subjects showed a fall in heart rate, skin temperature and core temperature
- The majority of these changes occur within 7-10 days of the onset of exposures to heat, and they last several weeks after the exposure
(Wyndham, 1968):
- Carried out a similar experiment and showed that sweat rate also increases.

What is required for rapid acclimation to heat?
Periods of exercise (90-120 min) are required for rapid acclimation, and the exercise is much better tolerated after acclimation.
Describe how sweating changes during heat acclimation.
The shift to the left is due to central acclimation (lower threshold for sweating), while the increased gradient is due to peripheral acclimation (greater sensitivity).

Describe how heatstroke occurs.
(Epstein, 2019):
- The sequence of events that leads to heat stroke involves a transition from a compensable thermoregulatory state to the noncompensable condition.
- Heat stress initiates a thermoregulatory response to increased cardiac output and redistribution of blood flow.
- When central venous pressure begins to decrease substantially, core temperature begins to increase rapidly and becomes noncompensable.
- The thermoregulatory failure aggravates pathophysiological processes at the cellular level, including an inflammatory reaction, and multiorgan failure occurs as a result of the combination of high body temperature and circulatory collapse, and ultimately is expressed as heatstroke.

Describe what happens to HIF in the presence/absence of oxygen.
- In the presence of oxygen, it is hydroxylated and degraded
- In the absence of oxygen, this does not happen

What are 3 physiological variations at high altitude? Are they advantageous or disadvantageous?
- Erythrocytosis -> Advantageous (unless it is excessive, causing chronic mountain sickness)
- Pulmonary arterial vasoconstriction -> Maladaptive
- Ventilatory acclimatisation -> Advantageous
Describe how haematocrit affects viscosity and cardiac work.
- Viscosity increases as haematocrit increases.
- Cardiac work is relatively unaffected because the arterioles can dilate in compensation for the increased pressure.

Give some experimental evidence for erythrocytosis being an advantage.
(Juvonen, 1991):
- Described a family with autosomal dominant erythrocytosis.
- In in vitro cultures, performed using the methyl cellulose assay, the number of erythroid colonies was normal or marginally increased when a standard concentration of erythropoietin (Epo) was used, but at lower Epo concentrations, the investigated persons formed more colonies than the controls. The difference was generally greater the lower the Epo concentration became.
- This finding indicates that the erythrocytosis in this family is caused by hypersensitivity of erythroid progenitors to Epo.
- Many of the family reached an advanced age, and one of the affected family members has won several Olympic gold medals and world championships in endurance sports.
Is our normal haematocrit optimal for exercise performance?
(Schuler, 2010):
- The optimum haematocrit in mice is higher than the average normal haematocrit

Are high altitude animals adapted to altitude by having high haematocrit? Give some experimental evidence.
(Hall, 1937):
- High-altitude mammals do not have increased oxygen carrying capacity (i.e. haematocrit)

Are high-altitude human populations adapted to altitude by having high haematocrit? Give some experimental evidence.
(Beall, 1998):
- Aymara have high haemoglobin concentrations, but Tibetans do not

What genetic adaptation allows Tibetans to be adapted to high altitudes?
- Tibetans have a high allele frequency of a certain allele for the EPAS1 gene
- This gene encodes HIF2α
- The allele is associated with low haemoglobin concentration, which explains why Tibetans tend to have similar haemoglobin concentrations to sea level populations
- However, the allele is also associated with improved oxygen transport

Give some experimental evidence for the role of the kidneys in producing EPO.
(Jacobson, 1957):
- Stimulated erythropoeisis by either administering cobalt or causing bleeding in animals
- Animals with their kidneys removed produced a smaller response (in terms of EPO production)

Explain why the kidney is a good location for EPO production.
- The kidney is a good place for EPO production because PO2 in the kidney is dependent on haematocrit, not blood flow
- In other organs, if you increase blood flow, then PO2 will increase because oxygen delivery is increased while oxygen consumption is unchanged
- However, in the kidneys, most oxygen consumed is used for reabsorption of ions from the ultrafiltrate. If you increase blood flow, then oxygen delivery is increased but oxygen consumption is proportionally increased because more ultrafiltrate is produced and thus more energy is required to reabsorb the ions.
- Therefore, only haematocrit can alter PO2 in the kidneys since it affects oxygen delivery. This means that the kidneys are good at sensing a need for erythropoeisis.
How can pulmonary arterial pressure be measured?
- Swan Ganz catheter (in diagram)
- Echocardiography of the right atrium
- When the right ventricle contracts, there is a jet of blood that flows to the right atrium
- Echocardiography allows the velocity of this to be calculated
- Bernoulli’s equation can be used to calculate pulmonary arterial pressure

How does pulmonary vasoconstriction change with prolonged exposure to hypoxia (over several hours)? Give some experimental evidence.
(Dorrington, 1997):
- Hypoxia leads to a two-stage rise in pulmonary vascular resistance over the first 3 hours -> The slower second increase is due to HIF
- At the end of hypoxia there is a sharp drop in PVR followed by a gradual return to baseline -> This is evidence of some sensitization following hypoxia

What causes Chuvash polycythemia? Give a reference.
(Ang, 2002):
- Chuvash polycythemia is caused by homozygosity of an allele for VHL that causes loss of function
- This means there is reduced clearance of HIF, leading to erythrocytosis, etc.

Describe hypoxic pulmonary vasoconstriction in patients with Chuvash polycythemia.
The HPV is more pronounced due to the lack of HIF degradation seen in Chuvash polycythemia. Even in normoxic conditions, the pulmonary arterial pressure is high.

Describe hypoxic pulmonary vasoconstriction in Tibetans. Give some experimental evidence.
- Tibetans have a high allele frequency of a certain allele for the EPAS1 gene
- This gene encodes HIF2α
- The allele is associated with low haemoglobin concentration
(Petousi, 2014):
- Han Chinese and Tibetans had their pulmonary hypoxic response tested before and after exposure to hypoxia for 8 hours
- The Tibetans showed a lesser increase in pulmonary arterial pressure upon exposure to transient hypoxia -> This is because HPV is partly mediated by HIF in response to sustained hypoxia.

How does iron affect HPV? Give some experimental evidence.
(Smith, 2008):
- Measured pulmonary hypoxic response before and after exposure to hypoxia for 8 hours
- The group who received iron infusion prior to the 8 hours of hypoxia showed a smaller increase in pulmonary arterial pressure upon transient hypoxia compared to the group receiving saline infusion
- This is suggested to be because iron is involved in hydroxylating HIF prior to degradation, so it promotes HIF degradation and therefore reduces HPV
(Smith, 2009):
- Sea level residents were taken to altitude and after 3 days either a placebo or iron was administered
- The iron group showed a more marked and rapid fall in pulmonary arterial pressure

HPV is driven by…
HIF
What is ventilatory acclimatisation to high altitude?
- A progressive rise in ventilation and fall in PCO2 that occurs over a number of days of exposure to high altitude.
- It is associated with increased sensitivity to both CO2 and hypoxia.
Is ventilatory adaptation to altitude an adaptation to altitude?
Some theories suggest so, but it could just be viewed as a consequence of of mechanisms of regulation of ventilation at sea level.
Give some experimental evidence for the importance of peripheral chemoreceptors in ventilatory acclimatisation to altitude.
Peripheral chemoreceptor denervation leads to decreases in ventilation and increases in PCO2 in response to altitude.

Describe the two core theories of how ventilatory acclimatisation to altitude occurs.
Theory 1 -> pH-dependent:
- Peripheral chemoreceptors detect the fall in PO2 and there is compensatory hyperventilation
- This leads to a respiratory alkalosis
- In response, a compensatory metabolic acidosis (by excreting bicarbonate) develops, which is associated with a drop in pH in a certain body compartments
- This drives the increased ventilation
- A strength of this model is that ventilation is sensitive to acute changes in CO2 and pH
Theory 2 -> pH-independent:
- Ventilatory acclimatisation arises directly from the effects of PO2 and is not mediated through pH effects (e.g. it could work via HIF)
Do changes in pH in any body compartments drive adaptation to altitude? Give some experimental evidence.
- When someone travels to altitude, there is an immediate increase in pH (respiratory alkalosis).
- In the blood, bicarbonate falls (metabolic acidosis) along with PCO2 sufficiently to maintain pH but not to lower it to normal. Therefore, it cannot be a peripheral pH sensor (peripheral chemoreceptors) driving acclimatisation in response to the pH returning to normal.
- In the CSF, the bicarbonate falls more quickly so the pH returns to normal. Thus, this drives hyperventilation through the central chemoreceptors.
(Forster, 1975):
- HOWEVER, when looking at a larger number of individuals, this was not the case. The blood and CSF HCO3- fell approximately at the same rate.
- The pH of the compartments was not compensated sufficiently to drive acclimatisation in the form of hyperventilation.
Overall, the early results supported a pH-dependent model of ventilatory acclimatisation, but the later study supports a pH-independent model of ventilatory acclimatisation to altitude.

Describe Bisgard’s experiments and what they tell us about acclimatisation to altitude.
- Bisgard surgically removed one of a goat’s carotid bodies and re-plumbed the second one so that the type of blood supplying the carotid body could be controlled independently of the blood supplying the rest of the body
(Bisgard, 1985):
- Only the carotid body was given hypoxic blood, while the rest of the body was given normoxic blood.
- This was sufficient to drive ventilatory acclimatisation, showing the importance of the carotid bodies and that systemic hypoxia is not required for acclimatisation.
Other experiments:
- If the rest of the goat is given hypoxic blood, while the carotid body is given normoxic blood, there is no acclimatisation.
- If the gas the goat breathes is high in carbon dioxide, then respiratory alkalosis (via hyperventilation) is prevented. When then carotid body receives hypoxic blood under these conditions, acclimatisation still occurs, showing that alkalosis is not required for acclimatisation.
- If the stimulus at the carotid body is changed (e.g. to hypercapnia) or the carotid body is stimulated, this is insufficient to drive acclimatisation.
Overall, this suggests that local hypoxia at the carotid body is both sufficient and necessary for ventilatory adaptation to altitude.
Is hypocapnia required for ventilatory acclimatisation to altitude? Give some experimental evidence.
(Robbins, 1985):
- Used a chamber to maintain one group isocapnic at all times, while the other group was allowed to have changes in blood CO2
- Both groups were exposed to 8 hours of hypoxia and showed similar increases in ventilation
- This suggests that hypocapnia is not required for ventilatory acclimatisation
(Robbins, 1998):
- Carried out a similar protocol and then plotted the ventilation against PCO2 curves after acclimatisation
- The isocapnic and poikilocapnic showed similar changes in their graphs
Thus, this supports a pH-independent model of ventilatory acclimatisation.
Does HIF underlie ventilatory acclimatisation to altitude? Give some experimental evidence.
(Hodson, 2016):
- Studied mice
- Conditional inactivation of Phd2 enhances ventilatory response to hypoxia in mice (since Phd2 usually degrades HIF)
- Conditional or constitutive inactivation of HIF-2 alpha, but not HIF-1 alpha, abrogates ventilatory acclimation to hypoxia
Chuvash polycythemia:
- Patients with Chuvash polycythemia show higher increases in ventilation in response to acute hypoxia compared to normal patients
- These patients have increased HIF activity
Thus, HIF-2 alpha in particular is likely to underlie acclimatisation to altitude.
Compare ventilation in low-landers travelling to altitude compared with long-term residents living at high altitude.
Longer-term residents show:
- Higher values for alveolar PCO2
- Blunted ventilatory responses to hypoxia (Severinghaus, 1966)

Compare the chemoreflex in low-landers travelling to altitude compared with long-term residents living at high altitude.
(Velarde, 2003):
- Used dopamine and somatostatin to block the chemoreflex in loew-landers and those living at high altitude
- At high oxygen concentrations, ventilation is only minimally blunted
- At low oxygen concentrations, ventilation is blunted far more in the low-landers
- This suggests higher chemoreflex activity in the low-landers, although it is suggested that both groups show similar levels of chemoreflex at their respective normal oxygen concentrations (it is just that the long-term altitude residents are exposed to greater hypoxia at rest)

Is the blunted ventilatory response to hypoxia of long-term residents of high altitude irreversible?
- There is a claim that the blunting of ventilatory sensitivity to hypoxia by residence at high altitude is irreversible -> This suggestion is controversial
(Gamboa, 2002):
- Compared low-landers (graph 1) with high-altitude individuals living at low altitude (graphs 2 and 3)
- Exposed each group to hypoxia
- Both groups showed significant, similar initial response to hypoxia, but the high-altitude individuals showed a more rapid decline of this response during the hypoxia
- Thus, this partly supports the idea that blunted ventilatory response is irreversible. It also shows the situation is more nuanced.
(Rivera, 2003):
- When high-landers living at low altitude return to high altitude, they show similar acclimatisation as low-landers
- In other words, the ventilatory response is not permanently affected in this way

Give a summary comparison of acclimatized lowlander (VAL) with high-altitude native (HAN).
- Both VAL and HAN have reduced end-tidal PCO2 and elevated ventilation at high altitude, but more so for VAL than HAN.
- The underlying mechanisms very different:
- VAL has augmented peripheral chemoreflex response to acute hypoxia
- HAN has depressed peripheral chemoreflex response to acute hypoxia
- Only the sustained component (not initial components) of HAN’s response to hypoxia is irreversibly blunted.
- HAN re-exposure to hypoxia induces same acclimatization response as for lowlander.
See previous flashcards for more detail.
Does ventilatory acclimatisation only happen with large changes of altitude?
(Donoghue, 2005):
- Found that even very small changes in PO2 can alter the ventilatory chemoreflex sensitivity to hypoxia
- This suggests that ventilatory acclimatisation is not just a characteristic of ascent to altitude, but it can also happen with small changes in altitude near sea level

Describe the effects of carotid body denervation on response to CO2.
(Pan, 1998):
- Denervated the carotid bodies in a goat
- Found that the ventilatory response to CO2 decreased over the first 4 days and then slowly recovered to close to normal
- This shows that the central chemoreflex is under the control of the peripheral chemoreceptors -> The fact that the central chemoreceptors are unaffected is why the response can recover
(Fatemian, 2003):
- Studied patients with bilateral carotid body resection
- Found that bilateral carotid body resection in humans elevates alveolar PCO2 and suppresses ventilatory sensitivity to CO2
- Thus it is clear that bilateral resection leads to alterations in the ventilatory set point

Give a summary of pulmonary ventilation and hypoxia.
- Both ventilatory acclimatization to hypoxia and lifelong hypoxia alter the respiratory set point, and affect the acute chemoreflex sensitivities to hypoxia and CO2
- Carotid body resection has permanent effects of the ventilatory set point and the respiratory sensitivity to CO2, as well as removing the acute ventilatory response to hypoxia.
- Although not so important for short-term regulation of breathing as CO2, hypoxia may play a significant role in calibrating the ventilatory set point and chemoreflex sensitivities of the respiratory controller.
What corollaries may be draw from the idea that humans are a sea-level design that may ascend to altitude?
- Corollary I: hypoxia-sensing mechanisms exist to subserve functions at altitudes close to sea level – not sense reductions in environmental PO2
- Corollary II: to a substantial degree, it is a matter of chance whether physiological responses to the hypoxia of high altitude are beneficial or deleterious
- As such, it may be beneficial to suppress activation of hypoxia-sensing mechanisms such as HIF at high altitude (and potentially also in hypoxia-related disease).
Give a summary of the concept of the acute chemoreflex and acclimatisation to altitude.
- When acutely exposed to hypoxia, humans hyperventilate -> This is the acute chemoreflex
- Upon prolonged exposure to hypoxia (e.g. at altitude), humans adapt so that ventilation is increased in response to altitude
- This may happen by a pH-dependent system (where respiratory alkalosis is compensated by metabolic acidosis, so that the drop in pH in some compartment drives adaptation) or a pH-independent system (where low PO2 itself drives adaptation directly).
- Overall, the pH-independent model seems more likely since hypoxia at the carotid bodies is sufficient for acclimatisation, while hypocapnia is not required.
- On a graph of PaCO2 against ventilation, a shift to the left is seen and can be explained by increased sensitivity of the central chemoreceptors to CO2 and an increased gradient can be explained by increased sensitivity of the peripheral chemoreceptors to O2.
- It is likely that HIF-2 alpha drives acclimatisation to altitude.
Summarise the control of ventilation.
- Peripheral chemoreceptors (carotid body) -> Samples arterial blood for decreases in PO2 or to a lesser extent increases in pH and PCO2
- Central chemoreceptors (medullary) -> Sample CSF pH, which is dependent on blood PCO2 and HCO3-
These communicate with medullary respiratory neurons, which control breathing.

Compare the carotid sinus and carotid body.
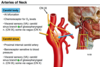
Draw a graph of ventilation against PCO2 and show how this graph changes during acclimatisation at high altitude.
- Parallel shift of line to the left (shown on the left graph)
- This is due to increased sensitivity of the central chemoreceptors to PCO2
- This change in sensitivity may be due to prolonged alkalinisation of the body as PCO2 falls
- The result is increased ventilation
- Increased slope of the line (shown on the right graph)
- This is due to an increase in the sensitivity of the peripheral chemoreceptors to PO2
- The result is increased ventilation
(Remember that this change only occurs over a few days as the body acclimatises to high altitude)

What may underlie the shift of the ventilation against PCO2 graph during acclimatisation to high altitude?

- It may be reduced CSF bicarbonate.
- This can be explained this way:
- At high altitude, there is hyperventilation
- This leads to falls in PCO2 in the body (respiratory alkalosis)
- The kidneys excrete increased amounts of bicarbonate to compensate for this
- So there are low levels of bicarbonate in the blood and CSF
Note that this explanation assumes a pH-dependent model of acclimatisation, which may not be the case.

Give some experimental evidence for the two types of shift that occur to the ventilation against PCO2 graph at altitude.
[EXTRA]

(Kellogg, 1963):
- Saw that the graph shifted to the left AND increased in slope
- Therefore, both types of shift occur

Give evidence for the importance of each of these mechanisms in acclimatisation to high altitude:
- Increased sensitivity of the central chemoreceptors to PCO2 (leading to a shift to the left on the ventilation-PCO2 graph)
- Increased sensitivity of the peripheral chemoreceptors to PO2 (leading to an increased slope on the ventilation-PCO2 graph)
[EXTRA]
Evidence that peripheral chemoreceptors detecting PO2 is important:
- (Howard, 1995) -> Varied PO2 without changing PCO2. It turns out that there is a large increase in ventilation upon hypoxia when PCO2 is kept constant.
- (Bisgard, 1986) -> Altered the blood supply to a goat’s brain so that the peripheral chemoreceptors can be independently perfused. Induced hypoxia at the carotid bodies but not at the central chemoreceptors. It turns out that ventilation increases when only the peripheral chemoreceptors are hypoxic.
Evidence that central chemoreceptors detecting PCO2 is important:
- (Ren, 1999) -> Asked patients to hyperventilate while keeping their PO2 constant. In patients where PCO2 was allowed to drop, the line shifted to the left. In patients where CO2 was supplied to keep PCO2 constant, the line did not shift.
Which is the more important mechanism in acclimatisation to high altitudes and why:
- Increased sensitivity of the central chemoreceptors to PCO2 (leading to a shift to the left on the ventilation-PCO2 graph)
- Increased sensitivity of the peripheral chemoreceptors to PO2 (leading to an increased slope on the ventilation-PCO2 graph)
- Increased sensitivity of the peripheral chemoreceptors to PO2 is likely to be the first response that kicks in, responsible for most of the acclimatisation.
- Increased sensitivity of the central chemoreceptors to PCO2 is likely to be slower because (in pH-dependent models) it relies on the kidneys excreting bicarbonate in order for CSF bicarbonate to fall.
Also note that it is likely that the peripheral chemoreceptors influence the central chemoreceptors.
Describe and explain the ventilatory response to altitude.
Assuming that there is a sudden change of altitude:
- PO2 drops and this hypoxia drives hyperventilation via the carotid bodies
- This increases PO2 and decreases PCO2
- The fall in PCO2 leads to alkalosis, which reduces ventilation via reduced stimulation of the central chemoreceptors
- This leads to a balance between hypoxia and hypocapnia
- Over several days, acclimatisation occurs, causing a shift towards hyperventilation

Summarise the main hypothesis as to why CSF bicarbonate falls during acclimatisation to high altitudes. Why is this important?
There are 3 main hypothesis:
- Renal compensation
- At high altitude, there is hyperventilation
- This leads to falls in PCO2 in the body (respiratory alkalosis)
- The kidneys excrete increased amounts of bicarbonate to compensate for this
- There is redistribution of bicarbonate from the CSF
- Active transport out of the CSF (not fully believed)
- At high altitude, there is hyperventilation, leading to respiratory alkalosis
- As pH of the CSF fluid falls, bicarbonate is actively transported out of it
- Lactacidosis
- At high altitudes, there is cerebral tissue hypoxia
- This leads to acidosis, which is buffered using bicarbonate

How would you approach a question about how acclimatisation to high altitude works?
- The classic assumption is a pH-dependent system of ventilatory acclimatisation, which requires bicarbonate excretion in order to happen
- The evidence for this includes, for example, the use of acetazolamide
- However, there are also a number of studies that point towards a pH-independent system that relies only of low PO2 to drive acclimatisation -> For example, bicarbonate excretion does not appear to happen rapidly enough to drive pH changes
- Overall, what we do know is the importance of the peripheral chemoreceptors in driving adaptation, followed by the central chemoreceptors
- This can be seen in the shape of the PaCO2-ventilation graphs
What is acute mountain sickness and what are the risk factors?
- Sickness that occurs during an ascent, typically before you acclimatise
- Risk factors include:
- Speed of ascent
- Final altitude
- Individual predisposition
What scale is used to assess acute mountain sickness? How is AMS defined?
- Lake Louise Score
- Defined as headache and Lake Louise Score >3, in setting of recent ascent/hypoxia (>6h)

What is the incidence of acute mountain sickness? Give some experimental evidence. Critique this evidence.
(Hackett, 1976):
- Trekkers were given a self-reported questionnaire in Pheriche (on Everest) during the return trek from base camp
- Incidence of AMS was 52.5% in 278 trekkers
- However, 20% of the subjects turned back on the way to Pheriche and some did not complete the questionnaire. Thus, AMS is likely to be under-reported here.
- The AMS questionnaire was before the Lake Louise score system was created, so the definition of AMS is not standardised here.
What may underlie altitude sickness?
- The cause is unclear but it is likely to involve a small degree of cerebral oedema.
- (Berger, 2020) found 3 main causes of AMS:
- Increased cerebral blood flow (due to vasodilation)
- Cytotoxic oedema (extracellular water passes into cells, resulting in their swelling)
- Vasogenic oedema (due to increased vascular permeability)
- These factors lead to increased brain volume and intravascular pressure that activate the trigeminal nerve

What are some possible explanations for people’s different susceptibilities to acute mountain sickness?
- There are some suggestions that it may depend on the amount of space that individuals have around the brain for the brain to expand into
- Also, there are factors that may affect the hypoxaemia experienced (and therefore the degree of AMS):
- Metabolic demand (e.g. exertion)
- Hypoxic ventilatory response
- Pulmonary oedema
Can we predict a person’s susceptibility of acute mountain sickness?
- It is difficult to predict.
- (Hohenhaus, 1995) studied the ventilatory response to isocapnic and poikilocapnic hypoxia and calculated the ventilatory sensitivity to hypoxia. The tests lasted 7-10 min, and were terminated when the SpO2 reached 80%. This seemed to identify those known to be susceptible to high altitude pulmonary oedema, but not AMS.
- (Burtscher, 2008) had a bit more success. They measured SpO2 after 20-30 min of hypoxia and had slightly more success in predicting who developed AMS.
- The best predictor seems to be previous experience of AMS.
How can acute mountain sickness be prevented? Give some experimental evidence for the best dose.
(Kayser, 2012):
- Reviewed 24 studies of acetazolamide in preventing AMS
- 250mg, 500mg and 750mg doses were all effective in preventing AMS
- However, high doses tend to have more side effects
A recent meta-analysis suggests 125mg twice daily is effective, with fewer side effects than higher doses. A more recent RCT suggests that 62.5mg twice daily is not effective (Lipman, 2021).
How does HAPE relate to acute mountain sickness?
HAPE often co-exists with AMS and HACE because it drives hypoxaemia, which is a driver of AMS and HACE.
What is high altitude cerebral oedema (HACE)? What are the causes, symptoms and treatment?
- Severe swelling of the brain due to fluid
Causes:
- Hypoxia leads to increased cerebral blood flow, increased cerebral blood volume and increased permeability of the BBB
Symptoms:
- Headache, malaise, ataxia, confusion and coma
Treatment:
- Descent
- Decompression
- Oxygen
- Dexamethasone
What is the difference between HACE and acute mountain sickness?
- HACE is a more severe form of AMS
- HACE includes confusion, ataxia and depressed consciousness (coma), making it more dangerous
Give some experimental evidence for what causes HACE.
(Hackett, 1998):
- Used MRI to scan an individual with HACE before and after recovery
- The before scan showed a high degree of vasogenic oedema, particularly in the corpus callosum
(Kellenberg, 2008):
- Found evidence of microbleeds (hemosiderin deposits) on MRI scans of patients recovering from HACE
- This evidence was not found in controls who went to similar altitudes and had severe AMS
- This gives radiological evidence for presence of cytotoxic as well as vasogenic oedema
Describe the treatment of HACE.
- Best treatment is descent, which is rapidly effective, but may be difficult
- If descent is not possible, oxygen or hyperbaria (using a Gamow bag) may be effective as holding measures
- The most important pharmacological treatment is dexamethasone (but acetazolamide may also be given)
What is high altitude pulmonary oedema (HAPE)? What are the causes, symptoms and treatment?
- Severe swelling of the lungs due to fluid accumulation
Causes:
- Widespread pulmonary vasoconstriction upon ascent to high altitudes
Strong individual predisposition
- Leading cause of death at high altitudes
Symptoms:
- Dry cough, then pink frothy sputum and progressive hypoxia
Treatment:
- Descent
- Oxygen
- Nifedipine -> Calcium channel blocker that reduces vasoconstriction
Describe how HAPE occurs.
- At altitude, there is some uneven hypoxic pulmonary vasoconstriction (HPV).
- This can lead to capillary pressures in some areas that are high enough to damage to capillary walls.
- Thus there is oedema in these parts of the lungs.
- (West, 1992) suggested this idea of stress failure first.

Describe the pulmonary oedema in HAPE.
It is patchy, unlike the oedema in heart failure.
How are HPV and HAPE related? Give some experimental evidence.
(Grunig, 1997):
- HAPE-susceptible volunteers have exaggerated HPV at sea level, suggesting that high pulmonary pressure may predispose to oedema at altitude

Give some experimental evidence for stress failure contributing to high-altitude pulmonary oedema (HAPE).
(Swenson, 2012):
- Flushed the lungs of healthy and HAPE-prone subjects with a fluid
- Measured the albumin concentration in this fluid, as well as the pulmonary arterial pressure
- As expected, those who were most severely ill had the highest pulmonary arterial pressure and also had high levels of albumin in the fluid
- This albumin is evidence of stress failure of capillaries causing the HAPE

Compare the pathology of HAPE and HACE.
- HACE is thought to be an inflammatory process that disrupts the BBB (secondary to the hypoxaemia of altitude)
- HAPE is thought to be due to stress failure of capillaries during uneven HPV (hypoxic pulmonary vasoconstriction)
These differences are evidenced by the presence or absence of inflammatory cells in the alveoli/brains of patients.
Give an example of a molecule that is upregulated in HAPE.
(Sartori, 1999):
- Compared 16 climbers prone to HAPE with 16 climbers resistant to HAPE at 4556m
- In the 16 susceptible climbers, endothelin-1 levels were significantly elevated
- Endothelin-1 is a potent vasoconstrictor released by the endothlium

Give an example of an uncommon drug that could be used to treat HAPE.
- Inhaled beta-2 agonists (i.e. salmeterol) reduce HAPE in susceptible individuals, perhaps by enhancing alveolar fluid clearance.
- Intravenous iron infusion rapidly reduces pulmonary artery pressure at high altitude. This is in keeping with a role for hypoxia-inducible factor in the pulmonary vascular response to high altitude.

What are some examples of sub-acute and chronic illnesses of high altitude?
- High altitude pulmonary hypertension (subacute mountain sickness)
- Chronic mountain sickness
What is high altitude pulmonary hypertension? What causes it and what does it lead to?
- High altitude pulmonary hypertension involves remodelling of the pulmonary vasculature such that the vessels are narrowed and there is pulmonary hypertension
- It is a chronic condition caused by chronic hypoxia
- The hypertension can lead to right sided heart failure and enlargement
What is an animal model for high altitude pulmonary hypertension?
Brisket disease:
- Lowland cattle taken to altitude in the early 1900s often died, with dependent oedema and poor exercise tolerance
- This was not acute, but took a while to happen
- Invasive measurements of pulmonary artery pressure revealed a progressive rise over weeks at altitude
- The rise in pressure seems almost universal, but varies considerably in magnitude
- In some cases it is severe enough to cause right heart failure (i.e. cor pulmonale)
Can subacute mountain sickness (high altitude pulmonary hypertension) be acutely reversed?
Right heart failure at altitude does not reverse acutely with oxygen, but does resolve slowly after weeks at sea level.
Give some experimental evidence for the mechanism of sub-acute mountain sickness.
(Groves, 1987):
- The pulmonary vascular pressure gradient normally increases slightly as cardiac output increases
- Studied 8 male volunteers by decompressing them in a hypobaric chamber for 40 days to a barometric pressure (PB) of 240 Torr, equivalent to the summit of Mt. Everest.
- Took haemodynamic measurements at the equivalents of 3 heights.
- With altitude, the relationship between cardiac output and pulmonary pressure steepens.
- This suggests a progressively less distensible pulmonary circulation, which provides evidence that sub-acute mountain sickness is caused by vascular remodelling that leads to pulmonary hypertension.
How can high altitude pulmonary hypertension (and right ventricular hypertrophy) be prevented in animal models?
- Endothelin antagonists (Chen, 1995)
- Inactivationof HIF
How does high altitude pulmonary hypertension (subacute mountain sickness) relate to HAPE?
- HAPE is an acute event, while high altitude pulmonary hypertension is subacute (since it takes some time for vascular remodelling to occur).
- However, both conditions are related and feature hypertension since HAPE involves uneven HPV that raises pulmonary pressure.
What is chronic mountain sickness and what is the mechanism?
- A condition seen in people living at high altitude for a long time, characterised by symptoms of headache, dizziness, fatigue and paraesthesia. There is also often severe pulmonary hypertension.
- The mechanism involves excessive erythropoiesis such that the viscosity of the blood is increased.
How is chronic mountain sickness diagnosed?
Diagnosis is based upon elevated haemoglobin (≥19 g/dL in females, ≥21 g/dL in males) and severe hypoxaemia in high altitude natives (>2500 m).
Describe what causes the excessive erythropoiesis in chronic mountain sickness. Give some experimental evidence.
(Monge, 1987):
- CMS is classically thought to be due to a loss of ventilatory adaptation to high altitude, such that hypoxaemia develops and so there is secondary erythrocytosis and pulmonary hypertension
- When plotting haematocrit against ventilation, there is an inverse relationship between the two.
- Patients with CMS are those with the lowest ventilation and highest haematocrit, suggesting that the erythocytosis is secondary to low ventilation.
(Bermudez, 2020):
- More recent data suggests that CMS may actually be the result of differences in gene expression that affect erythropoiesis.
- Under normoxic conditions, SENP1 expression is equally low in both CMS patients and non-CMS patients
- Under hypoxic conditions, SENP1 expression is very elevated, but only in CMS patients

How can chronic mountain sickness be treated?
- Intermittent or permanent relocation to lower altitude is effective at limiting erythrocytosis, but may be very difficult.
- Venesection produces symptomatic benefit but the effects may be shortlived (and iron deficiency could exacerbate pulmonary hypertension)
- Acetazolamide seems to produce improvements in oxygenation and haemoglobin in high altitude residents wit CMS (Richalet, 2008)
Summarise the difference between:
- Acute mountain sickness
- HACE
- HAPE
- Subacute mountain sickness
- Chronic mountain sickness
- Acute mountain sickness -> A mild form illness upon first ascent to altitude, thought to be largerly due to changes in cerebral blood flow and a small degree of cerebral oedema
- HACE -> A more severe form of acute mountain sickness, with more severe cerebral oedema. Thought to involve inflammation.
- HAPE -> Oedema of the lungs, involving stress failure of capillaries due to uneven hypoxic pulmonary vasoconstriction (rather than an inflammtory process).
- Subacute mountain sickness -> A.K.A. High altitude pulmonary hypertension. Due to vascular remodelling causing the pulmonary vessels to become less compliant.
- Chronic mountain sickness -> Chronic condition that involves excessive erythopoiesis in hypoxic conditions.
Describe two populations with different prevalences of chronic mountain sickness. Give some experimental evidence.
- Tibetans have a very low prevalence of CMS, due to a high allele frequency of a certain allele for the EPAS1 gene, which encodes HIF-2α (Beall, 2010). The allele is associated with low haemoglobin concentration compared to the Han Chinese, but the allele is also associated with improved oxygen transport. These results suggest a degree of selection against HIF-driven erythropoiesis due to the high risk of chronic mountain sickness, although there is a possibility that selection happened against another aspect of the human phenotype that EPAS1 determines.
- On the other hand, Andeans tend to show significantly greater erythropoiesis, which is associated with chronic mountain sickness.
For this question, name some advantages of doing the study in a lab or on a mountain:
Can intravenous iron reverse hypoxic pulmonary vasoconstriction?
In a lab:
- Controlled conditions
- Safe for participants and investigators
- Easy troubleshooting
- Predictability
On a mountain:
- Time efficiency
- Local populations
- International collaboration
- Exciting for researchers
- High impact
Describe the study (Beall, 2010).
- Carried out a genome-wide allelic differentiation scan to compare Tibetans with Han Chinese
- 8 SNPs achieved genome-wide significance close to EPAS1
- There was strong linkage disequilibrium between these SNPs and the SNPs within EPAS1 (i.e. these SNPs are co-inherited with EPAS1 because they are close to it on the chromosome)
- These 8 SNPs formed an extended haplotype (set of a genes inherited together from a single parent) that had a frequency of 46% in the Tibetans but only 2% in the Han Chinese
- This explains why Tibetans have a very low prevalence of CMS, due to a high allele frequency of a certain allele for the EPAS1 gene, which encodes HIF-2α
- The allele is associated with low haemoglobin concentration compared to the Han Chinese, but the allele is also associated with improved oxygen transport.
- Homozygotes for this allele have haemoglobin around 1g/dL than heterozygotes.
Describe the study (Simonson, 2010).
- Aimed to find genes that could be related to Tibetans’ adaptation to high altitude
- First, identified candidate genes by selecting genes with a known function that could be related to high altitude adaptation
- Secondly, a genome-wide scan was conducted to identify regions that show strong evidence of local positive selection in high-altitude Tibetans
ASK AND DD INFORMATION

Name three adaptations that make Tibetans a unique high-altitude population.
- Low haemoglobin concentration (Beall, 1998)
- High birth weight at altitude (Moore, 2001) -> There is usually a fall in birthweight with increasing altitude, but Tibetans experience a much smaller fall compared to Han Chinese
- Low prevalence of chronic mountain sickness (Moore, 2001)
- Low HPV (Groves, 1993)
- High hypoxicventilatory response
Summarise the response of Tibetans to hypoxia.
- They have blunted physiological responses to hypoxia (due to genetic variation in the HIF pathway)
- This is contrasted by patients at sea level with mutations in the HIF pathway that lead to exaggerated responses to hypoxia
Describe the genetics of EPAS1 in Tibetans compared to Han Chinese that lead to adaptation to high altitude. Give experimental evidence.
(Yi, 2010):
- Sequenced 50 exomes
- Did not find any significant SNPs in the exos
- However, they did find 2 highly divergent SNPs in the introns (very close to the exons which is why they were picked up) close to the EPAS1 gene
- These SNPs could have an effect by being parts of promoter regions, etc.
Describe the genetics of EGLN1 in Tibetans that lead to adaptation to high altitude. Give experimental evidence.
(Petousi, 2014):
- Found two SNPs in exon 1 of EGLN1 that are found in high frequency in Tibetans -> The two SNP frequently co-occur
- These variants are found in lower frequency in other Asians and are rare in Caucasians
- Also studied the relationship between the genotype and phenotype
- Found that those homozygous for the variant showed a more blunted erythropoietin response to hypoxia
- However, studies of Hb at altitude showed mixed results
(Lorenzo, 2014):
- Found that the frequency of these variants in Tibetans increases with altitude

Compare how EPAS1 and EGLN1 mutations enable adaptation to altitude in Tibetans.
- EPAS1 variants are in the introns -> This affects promoter regions, etc.
- EGLN1 variants are in the exons -> This affects the actual structure of the protein
What does EGLN1 encode?
PHD2
What does EPAS1 encode?
HIF-2α
Is the EGLN1 mutation in Tibetans gain of function or loss of function? Give experimental evidence.
- EGLN1 encodes PHD2, so logically it would make sense for it to be a gain of function mutation, since this would lead to reduced accumulation of HIF -> This would explain the reduced response to hypoxia seen in Tibetans (except the increased ventilation)
- (Lorenzo, 2014):
- Showed that the existence of a gain of function mutation in EGLN1 in Tibetans
- However, there is some contradictory evidence.
- (Song, 2020):
- Created a mouse model for the Tibetan EGLN1 mutation.
- Found that right ventricular systolic pressure (a measure of pulmonary hypertension) was not affected by the EGLN1 (PHD2) mutation, but the EPAS1 (HIF2a) mutation (diagram B).
- The EGLN1 mutation mice showed a higher hypoxic ventilatory response (diagram A) which suggests a loss of function mutation in the EGLN1 (PHD2).
See next flashcard if unsure.

Explain why Tibetans shown seemingly contradictory high hypoxic ventilatory response, as well as blunted pulmonary hypertension and blunted haemoglobin.
- It would seem that all of these effects should be either elevated or blunted since they are all under the control of the HIF pathway.
- (Song, 2020):
- Created a mouse model for the Tibetan EGLN1 mutation and compared it to a wild-type mouse.
- Found that right ventricular systolic pressure (a measure of pulmonary hypertension) was not affected by the EGLN1 (PHD2) mutation, but the HIF2a mutation (diagram B).
- The EGLN1 mutation mice showed a higher hypoxic ventilatory response (diagram A).
- These results suggests that Tibetans have a loss of function mutation in ELGN1 (PHD2) leading to an elevated hypoxic ventilatory response and a loss of function mutation in EPAS1 (HIF2a) leading to reduced pulmonary hypertension and blunted haemoglobin.

Summarise the cascade of events following immersion in water.
- Whole body immersion exerts pressure that compresses the limbs -> Increases venous return and central blood volume by around 700 ml
- There is also decreased lung volume by compression (less so when using SCUBA, since the air is delivered at ambient pressure)
- Right atrial and pulmonary arterial pressure increased by 15-20 mmHg
- Stroke volume and cardiac output by up to 30%
- The increased blood pressure leads to increased natriuresis and diuresis
- The increased blood pressure is further exacerbated by cold-induced vasoconstriction
Describe how fluid movement in the lungs is affected during immersion in water.
- Hydrostatic pressure at the start of the pulmonary arteries is increased (due to compression of the limbs and peripheral vasoconstriction causing increased central volume)
- But the colloid pressure throughout the arteries is unchanged
- Overall, Starling forces mean that there is a net outwards force that causes fluid movement out of the vessels and into the alveoli
- This pleural effusion leads to stiffer lungs, increased pulmonary vascular resistance and increased pulmonary arterial pressure
- Eventually, there is pleural oedema, leading to reduced surface for gas exchange

What can increase the incidence of immersion pulmonary oedema and why?
- Certain breathing systems can increase the work required to breathe, resulting in a low pressure in the alveoli
- This low pressure affects Starling forces, causing more fluid to be dragged out from the blood vessels
- For example, a back-mounted closed-circuit rebreather can involve the air bag being at higher pressure than the lungs
- Over 40 years, 11 cases of IPO have been recorded in French navy divers, probably due to negative-pressure breathing

What drugs have been shown to alleviate immersion pulmonary oedema?
- Nifedipine
- Sildenafil
Draw a diagram of the main contributing factors to IPO (immersion pulmonary oedema) and list them.
- Hyper-hydration
- Exertion
- Cold-induced vasoconstriction
- Hypertension
- Increased gas density at depth
- Work of breathing

How much does pressure increase with depth when diving?
For every 10m, the pressure increases by 1 atmosphere.
What is Boyle’s Law?
P1V1 = P2V2
What is Henry’s Law?
- The amount of gas that will dissolve in a liquid is almost directly proportional to the partial pressure of that gas.
- It is time-dependent and temperature-dependent, where higher temperature makes the gas less soluble.

Does lung volume change while diving?
No, because the diver is breathing air at ambient pressure.
Describe dissolved gas fluxes as you dive to a depth and then return to the surface.
- At the surface, your tissues are in equilibrium with the environment (i.e. your lungs)
- As you descend, the partial presssure of the gases in your lungs increases, driving fluxes towards tissues
- A new equilibirum is eventually established
- Upon return to the surface, the fluxes are reversed

Describe how the gas saturation of body tissues changes with time spent underwater.
- Gas saturation changes exponentially with time (since gas fluxes take time to happen)
- The body can be divided into several compartments (e.g. the Buhlmann ZH 16 algorithm) based on how long it takes for that tissue to become saturated
- The nervous system and spine get saturated very fast (fast tissues), whereas fat and bonessaturate very slowly (slow tissues)

What is inert gas narcosis?
- When diving, the body’s tissues become saturated with high partial pressure of gases being breathed in (including oxygen and nitrogen) since these are administered at ambient pressure
- Many gases are mild anaesthetics and the effects are directly related to partial pressure
- Thus, inert gas narcosis includes effects similar to alcohol
- The mechanism is not known but is thought to maybe involve disruption of the lipid bilayer by the gas

Summarise the effects of nitrogen narcosis at different depths.

Give some experimental evidence for the effects of nitrogen narcosis.
- Subjects were exposed to gas at 1 atm and then at 6 atm
- The average reaction time for the Stroop test was over 2 seconds slower at 6 atm and the accuracy was far worse
- This shows that executive functions are impaired by inert gas narcosis
[FIND REFERENCE]

What can make inert gas narcosis better or worse?
- Acclimatisation can occur with subsequent dives
- Effect is increased when PCO2 is high (e.g. when exercising)
Describe what oxygen toxicity is when diving.
Lorrain Smith effect (Pulmonary oxygen toxicity):
- Pulmonary oxygen toxicity can occur with piO2 values as low as 0.5 ATA if the exposure is sufficiently long
- Can be found in ICU and hyperbaric medicine units
- It involves damage to the lung epithelium and lung oedema (fibrosis etc.) since there is a lack of antioxidants -> This results in hypoxaemia
Paul Bert effect (CNS oxygen toxicity):
- A grand mal seizure can occur if a subject breath oxygen at 3 ATA but can also occur at pO2 of 2 ATA (at rest) for a prolonged period
- Signs of acute O2 toxicity: visual disturbances, ringing in the ears, dizziness, mood swings, convulsions, coma
Give some experimental evidence that hyperbaric oxygen can lead to pulmonary inflammation.
(Demchenko, 2007):
- Studied the bronchoalveolar lavage fluid from rats after exposure to normobaric or hyperbaric oxygen.
- The number of inflammatory cells increased gradually over 56hrs in the group at 1 ATA oxygen and much faster in the groups at 2, 2.5 and 3 ATA
- Lactate dehydrogenase is a marker of cell damage
- Similar trends of lactate dehydrogenase concentration were seen as with the inflammatory cells

What are the two main stages of pulmonary inflammation due to hyperbaric oxygen (when diving to a depth)?
Exsudative stage:
- There is pulmonary oedema, with breakdown of the pulmonary epithelium and an inflammatory infiltrate
Proliferative stage:
- Occurs when the exudative stage resolves and there is fibrotic tissue
Overall this means that the work of breathing increases and there is hypoxaemia.
Describe the mechanism of acute oxygen toxicity.
- Reactive oxygen species (produced due to high partial pressures of oxygen) lead to:
- Membrane disruption / inactivation of cell membrane transport
- Protein / enzyme inactivation
- Impaired electron transport
- DNA and RNA damage
- ROS react with NO to produce ONOO- (peroxynitrite) that reacts with protein tyrosines to nitrate them -> This is thought to be the cause of seizures (Chavko, 2003)
- This can lead to lipid peroxidation and GABA metabolism is disrupted, contributing to seizure onset
Give some experimental evidence for the importance of protein nitration in the onset of hyperoxic seizures.
(Chavko, 2003):
- Used a Western blot to measure the number of nitrated tyrosines over time with exposure to hyperbaric oxygen
- After exposures, brains were analyzed for protein nitrotyrosine (NT), with NT concentration increasing with time
- An NOS inhibitor can be used to block this effect

How can the dangers of inert gas narcosis be avoided?
Often a gas mixture is used that has less N2 and O2, helping to avoid the toxicity of these gases.
What are some different types of diving gases that may be used?
- Mixtures containing lots of O2 and N2 may only be used near the surface and at slight depths
- Mixtures with less O2 and N2 may be used at greater depths but not near the surface so a second gas mixture must be carried

What are barotraumas and what are some common locations?
Barotraumas are injuries that are caused by changes in pressure in certain body compartments.

What is a common barotrauma and what causes it? What are the symptoms?
- Pulmonary over-inflation barotrauma is the second most common cause of death in divers aside from drowning
- Caused by:
- Breath-holding during ascent
- Uncontrolled ascent
- Inhaling while pushing the “purge” button
- Air trapped in the lung
- Symptoms:
- Most (95%) cases show varying systemic neurologic signs but vital signs preserved
- Few (5%) cases show apnea, unconsciousness and cardiac arrest
What happens at tissues saturated with gases when you return to the surface after a dive?
Decompression from saturation is associated with formation of inert gas bubbles (usually nitrogen) in the tissue and veins (venous gas emboli, VGE).
How do gas bubbles form during decompression from diving?
There are multiple theories of bubble formation, including: microbubbles/micronuclei (little direct evidence), crevice model, tribonucleation and differential bubble seeding depending upon substrate (e.g. fat/muscle)
Why do decompression bubbles form largely in veins?
There is less oxygen in veins so there is a large nitrogen gradient for the formation of N2 bubbles.
How can decompression bubbles be detected?
Bubbles can be detected using Doppler ultrasound.
How does the rate of decompression affect bubble formation and the effect of adverse effects?
(Zhang, 2016):
- Found that the numbers of bubbles correlate with decompression rate
- The number of bubbles peaked about 20 minutes after decompression
- The 3 minute ascent peaked at a bubble score of 4, while the 5 minute ascent peaked at a bubble score of 2 on average
(Papadopoulou, 2015):
- The higher the bubble grade, the high the risk of adverse effects

Is venous gas emboli formation upon decompression variable?
- Yes, significant inter-personal variability is seen under the same controlled conditions
- Various factors can affect bubble formation:
- Exact dive profile
- Previous exposures
- Hydration
- Vascular health
- Thermal comfort
- Exercise/workload
- State of mind
- Silent bubbles
(Papadopoulou, 2018)

What are the effects of venous gas emboli (VGE) upon decompression?
- Activate innate immunity (complement)
- Damage the endothelium, activating endothelial markers of stress:
- There are increased levels of endothelin and decreased levels of NO
- Subsequently, there is activation of inflammatory pathways (↑TNF-a, IFN-g and IL-6)
Endothelial activation correlates with bubble number.

By what mechanism do VGE affect endothelial cells?
- Endothelial cells respond to microbubble contact via mechano-transduction responses
- Bubble contact stimulates extracellular calcium entry -> (Kobayashi, 2011) used fluorescence microscopy images to show that calcium entry occurs shortly after bubble contact
- Superoxide and peroxynitrite ions are formed in endothelial cells and are involve in the attenuation of NO and cell death
- Bubble contact and pressure change also drives the release of EMPs (Endothelial microparticles), which have a role in coagulation, inflammation, endothelial function, and angiogenesis
What are EMPs?
- Endothelial microparticles
- These are complex vesicular structures shed from endothelial cells (100 nm to 1 μm in diameter)
- Their release is driven by bubble contact and pressure changes
- They have a role in coagulation, inflammation, endothelial function, and angiogenesis
- They are proposed to be a cause of DVT during flying and can pass through the lugs and have secondary systemic effects
What happens to venous gas emboli?
- They are mostly filtered out by the lungs
- If there are too many bubbles (or under certain circumstances) these bubbles can bypass the lung filter and enter the arterial circulation
- Arterialisation of gas emboli can lead to symptoms of decompression sickness (DCS)
What are some ways of mitigating decompression sickness?
- Slow ascent
- Switching to breathing a gas mixture with more oxygen (rather than nitrogen) during ascent -> This encourages a lower partial pressure of gases in the venous circulation (since the oxygen is metabolised in tissues), so that nitrogen gas is dragged out from tissues and bubbles into the venous blood. This allows it to be breathed out.
- Exercise before diving
- Exercise during ascent (but NOT during descent)
- Avoiding exercise after diving
What is the oyxgen window?
- The difference between the partial pressure of oxygen in arterial blood and in body tissues.
- It is caused by metabolic consumption of oxygen.
How does exercise before diving affect VGE formation? Why?
- Aerobic exercise immediately before diving reduces VGE formation
- This is likely to be because exercise improves endothelial NO bioavailability
- This leads to:
- Improved endothelial health
- Less local sheer stress-induced inflammation
- Less bubble-mediated dysfunction
- NOS inhibition increases bubble formation in experimental models
[FIND REFERENCES]

How does exercise during diving affect VGE formation? Why?
- Exercise during compression:
- The raised cardiac output increase gas loading (so tissues are more saturated), narcosis severity (CO2) and O2 toxicity
- This increases the likelihood of VGE
- Exercise during decompression (ascent)
- Moderate swimming performed during a 3 min decompression stop reduces post-dive VGE and may reduce the number of nuclei from which bubbles form
[FIND REFERENCES]

How does exercise after diving affect VGE formation? Why?
- It can increase the risk of VGE
- This is because cardiac output may be increased to the extent that VGEs pass quickly through the lungs without being filtered out
- Thus, arterialisation occurs, which is the cause of decompression sickness
- (Eldridge, 2004):
- Studied 24 men who were subjected to exercise of progressive intensity, up to VO2max.
- Contrast echocardiography was used to detect the first % of the VO2max at which shunting occurred.
- In all cases, the intrapulmonary shunting developed at submaximal exercise levels.

Define decompression sickness and decompression illness.
- Decompression sickness (DCS) -> Caused by the formation of intra-corporeal bubbles resulting in a drop in pressure
- Decompression Illness (DCI) -> A combination of DCS and arterial gas embolism (AGE) which can be as a result of barotrauma, shunt or rapid decompression.
What are the symptoms of decompression sickness?
It depends on where the arterialised bubbles end up, but symptoms include:
- Joint pain
- Neurological symptoms
- Audio-vestibular symptoms
- Cutaneous symptoms
- Cardiopulmonary symptoms
- Lymphatic symptoms
- Girdle pain
- Constitutional
How is decompression sickness treated?
Hypobaric Oxygen Treatment:
- This is recompression with hyperbaric oxygen
- Essentially, the patient experiences a bubble-crushing ‘dive’ with slow and controlled decompression



