P2- Radioactivity Flashcards
(22 cards)
Why can’t we predict when radioactive decay will occur?
Radioactive decay is a random event and is not affected by external conditions.
What is background radiation?
Background radiation is from radioactive substances in the environment, or from space, or from devices such as X-ray machines.
What was Dalton’s theory on the atom?
1800s:
- All matter is made of atoms.
- Atoms cannot be broken down.
- Atoms of one element are identical.
- Compounds are formed when 2+ different atoms join together.

What was the ‘plum pudding model’?
Thomson- 1897:
Atoms consisted of spheres of positive charge with electrons stuck into them, like plums in a plum pudding.

Describe Rutherford’s experiment (1911).
Alpha particle scattering experiment➡️Fired alpha particles at thin gold foil.

What was the result of Rutherford’s experiment and how was this explained?
- Most particles passed straight through the foil➡️Most atom is empty space.
- Some particles deflected➡️Nucleus has positive charge.
- A few particles reflected➡️Nucleus has large mass and large positive charge.

What did Rutherford’s experiment prove?
The atom is mainly empty space, with a positive nucleus.

Define isotope.
An atom of the same element but with a different number of neutrons.
What is the relative charge of an alpha particle?

+2.
What is the relative charge of a beta particle?

-1.
How far is alpha’s penetrating power?
Paper or a few centimetres of air.

How far is beta’s penetrating power?
Thin metal (e.g: aluminium) or about a metre of air.
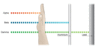
How far is gamma’s penetrating power?
Thick lead.

What can deflect alpha and beta particles?
Electric fields and magnetic fields.
Which type of nuclear radiation is the least ionising?
Gamma rays.
What is half-life?
The time required for half of the nuclei of a given amount of radioactive substance to disintegrate.
What has happened to the original count rate of a radioactive sample after two half-lives have passed?
The count rate is a 1/4 of the original.
A radioactive isotope produces a count rate of 800cpm.
After 8 minutes the count rate has decreased to 200cpm.
What is the half-life of the radioactive isotope?
800➡️400➡️200= 2 half-lives.
8/2= 4 minutes= half-life of radioactive isotope.
A radioactive isotope has a half-life of 6 hours.
It is injected into a persons body and the count rate at the start is 1600cpm.
What will the count rate be after 24 hours.
24/6= 4 half-lives.
1600➡️800➡️400➡️200➡️100.
100 counts per minute after 24 hours.
A radioactive substance has a half-life of 10 days.
If there is 64g of the radioactive substance now, how much was there 30 days ago?
30/10= 3 half-lives.
64➡️128➡️256➡️512.
30 days ago= 512g.
Carbon-14 has a half-life of 6000yrs.
A skeleton is found to have a count rate of 30cpm. A living person would have a count rate of 120cpm.
How old is the skeleton?
120➡️60➡️30= 2 half-lives.
6000 x 2= 12,000yrs old.
What are the three main types of radiation?
Alpha, beta and gamma radiation.


